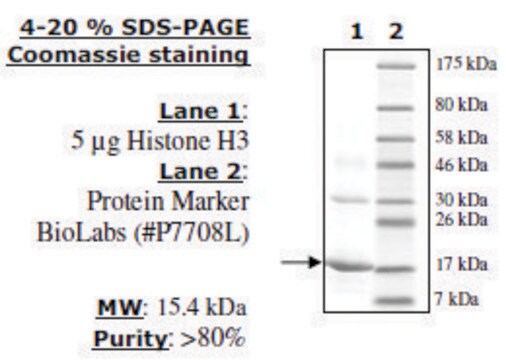
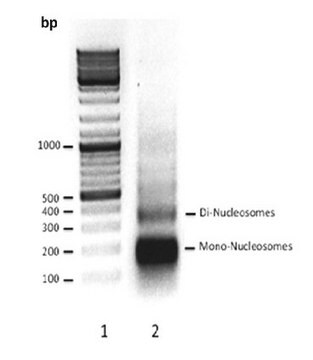
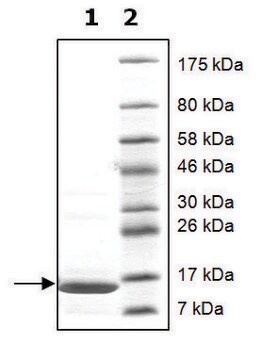
SRP0408
Histone Octamer full length human
recombinant, expressed in E. coli, ≥90% (SDS-PAGE)
Synonim(y):
Histone Octamer
About This Item
Polecane produkty
pochodzenie biologiczne
human
rekombinowane
expressed in E. coli
Próba
≥90% (SDS-PAGE)
Postać
aqueous solution
masa cząsteczkowa
113.8 kDa
opakowanie
pkg of 100 μg
metody
cell based assay: suitable
rozpuszczalność
water: soluble
Warunki transportu
dry ice
temp. przechowywania
−70°C
informacje o genach
human ... HIST1H2BG(8339) , HIST2H2AC(8338) , HIST3H3(8290) , HIST4H4(121504)
Opis ogólny
The histone octamer is a versatile protein assembly that has evolved to serve two opposing functions within the cell. It is required to bind and bend DNA to achieve fivefold compaction and partial charge neutralization of DNA, while also needing to release specific segments of DNA in a coordinated manner to allow the access of DNA-processing enzymes at the appropriate time. A modular assembly of histone dimers (consisting of either H2A and H2B or H3 and H4) binds to approximately 30 bp of DNA and is connected in a flexible yet stable manner to form a fundamental superhelical ′ramp′ with evenly spaced DNA-binding platforms.
Zastosowanie
Działania biochem./fizjol.
Kod klasy składowania
10 - Combustible liquids
Klasa zagrożenia wodnego (WGK)
WGK 1
Temperatura zapłonu (°F)
Not applicable
Temperatura zapłonu (°C)
Not applicable
Certyfikaty analizy (CoA)
Poszukaj Certyfikaty analizy (CoA), wpisując numer partii/serii produktów. Numery serii i partii można znaleźć na etykiecie produktu po słowach „seria” lub „partia”.
Masz już ten produkt?
Dokumenty związane z niedawno zakupionymi produktami zostały zamieszczone w Bibliotece dokumentów.
Klienci oglądali również te produkty
Nasz zespół naukowców ma doświadczenie we wszystkich obszarach badań, w tym w naukach przyrodniczych, materiałoznawstwie, syntezie chemicznej, chromatografii, analityce i wielu innych dziedzinach.
Skontaktuj się z zespołem ds. pomocy technicznej