Kluczowe dokumenty
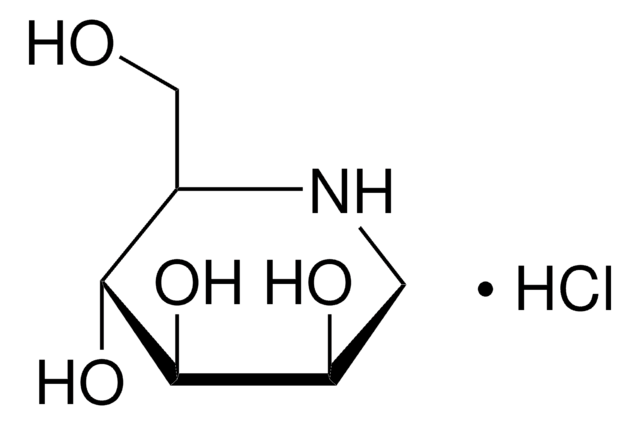
S8195
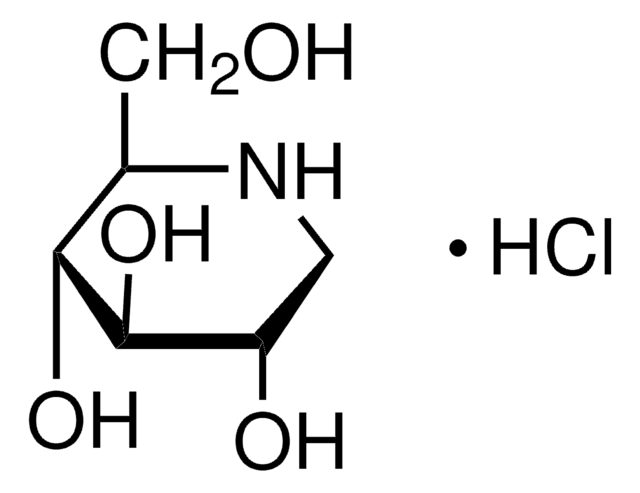
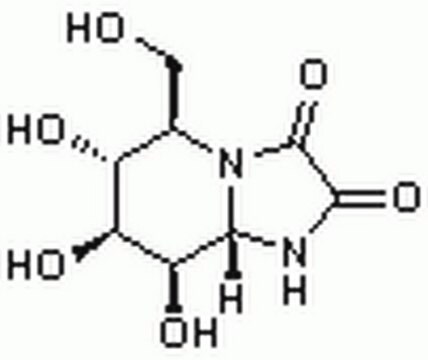
Swainsonine
from Metarrhizium anisopliae, ≥98% (TLC)
Synonim(y):
(1S,2R,8R,8aR)-1,2,8-Octahydroindolizidinetriol
About This Item
Polecane produkty
pochodzenie biologiczne
Metarrhizium anisopliae
Poziom jakości
Próba
≥98% (TLC)
Postać
lyophilized powder
warunki przechowywania
(Keep container tightly closed in a dry and well-ventilated place.)
kolor
white to faint yellow
rozpuszczalność
H2O: soluble 1 mg/mL
spektrum działania antybiotyku
neoplastics
Tryb działania
enzyme | inhibits
temp. przechowywania
2-8°C
ciąg SMILES
O[C@@H]1CCCN2C[C@@H](O)[C@@H](O)C12
InChI
1S/C8H15NO3/c10-5-2-1-3-9-4-6(11)8(12)7(5)9/h5-8,10-12H,1-4H2/t5-,6-,7?,8-/m1/s1
Klucz InChI
FXUAIOOAOAVCGD-DCDLSZRSSA-N
Szukasz podobnych produktów? Odwiedź Przewodnik dotyczący porównywania produktów
Powiązane kategorie
Zastosowanie
Działania biochem./fizjol.
Opakowanie
Uwaga dotycząca przygotowania
Inne uwagi
Kod klasy składowania
11 - Combustible Solids
Klasa zagrożenia wodnego (WGK)
WGK 3
Środki ochrony indywidualnej
dust mask type N95 (US), Eyeshields, Gloves
Certyfikaty analizy (CoA)
Poszukaj Certyfikaty analizy (CoA), wpisując numer partii/serii produktów. Numery serii i partii można znaleźć na etykiecie produktu po słowach „seria” lub „partia”.
Masz już ten produkt?
Dokumenty związane z niedawno zakupionymi produktami zostały zamieszczone w Bibliotece dokumentów.
Nasz zespół naukowców ma doświadczenie we wszystkich obszarach badań, w tym w naukach przyrodniczych, materiałoznawstwie, syntezie chemicznej, chromatografii, analityce i wielu innych dziedzinach.
Skontaktuj się z zespołem ds. pomocy technicznej