Wszystkie zdjęcia(1)
Kluczowe dokumenty
K3125
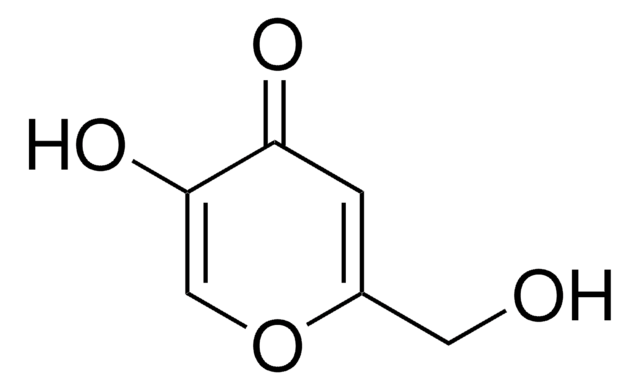
Kojic acid
≥98.5% (HPLC), powder, tyrosinase inhibitor
Synonim(y):
2-Hydroxymethyl-5-hydroxy-γ-pyrone, 5-Hydroxy-2-hydroxymethyl-4H-4-pyranone
Zaloguj sięWyświetlanie cen organizacyjnych i kontraktowych
About This Item
Wzór empiryczny (zapis Hilla):
C6H6O4
Numer CAS:
Masa cząsteczkowa:
142.11
Beilstein:
120895
Numer WE:
Numer MDL:
Kod UNSPSC:
12352106
Identyfikator substancji w PubChem:
NACRES:
NA.77
Polecane produkty
Nazwa produktu
Kojic acid,
Próba
≥98.5% (HPLC)
Formularz
powder
mp
152-155 °C (lit.)
ciąg SMILES
OCC1=CC(=O)C(O)=CO1
InChI
1S/C6H6O4/c7-2-4-1-5(8)6(9)3-10-4/h1,3,7,9H,2H2
Klucz InChI
BEJNERDRQOWKJM-UHFFFAOYSA-N
Szukasz podobnych produktów? Odwiedź Przewodnik dotyczący porównywania produktów
Zastosowanie
Kojic acid has been used:
- as an inhibitor of tyrosinase in guinea pigs pigmented hyperopic (PH)
- as a reference inhibitor standard for screening tyrosinase inhibition
- as a positive control for inhibition of tyrosinase in B16F10 melanoma cells
Działania biochem./fizjol.
Kojic acid is derived from some fungal species such as, Aspergillus, Acetobacter and Penicillium.. It halts melanin synthesis by inhibiting tyrosinase enzyme. It is used in the preparation of skin whitening cosmetics. However, kojic acid usage is minimal in cosmetics, as it induces skin irritation by its unstability and cytotoxic nature during long storage. It is an antioxidant and elicits radioprotective effects on chelating with manganese and zinc.
Tyrosinase inhibitor.
Ta strona może zawierać tekst przetłumaczony maszynowo.
Kod klasy składowania
11 - Combustible Solids
Klasa zagrożenia wodnego (WGK)
WGK 1
Temperatura zapłonu (°F)
Not applicable
Temperatura zapłonu (°C)
Not applicable
Środki ochrony indywidualnej
Eyeshields, Gloves, type N95 (US)
Wybierz jedną z najnowszych wersji:
Masz już ten produkt?
Dokumenty związane z niedawno zakupionymi produktami zostały zamieszczone w Bibliotece dokumentów.
Klienci oglądali również te produkty
Effects of the Tyrosinase-Dependent Dopaminergic System on Refractive Error Development in Guinea Pigs
Jiang L, et al.
Investigative Ophthalmology & Visual Science, 59(11), 4631-4638 (2018)
Ka-Heng Lee et al.
European journal of medicinal chemistry, 44(8), 3195-3200 (2009-04-11)
A series of 46 curcumin related diarylpentanoid analogues were synthesized and evaluated for their anti-inflammatory, antioxidant and anti-tyrosinase activities. Among these compounds 2, 13 and 33 exhibited potent NO inhibitory effect on IFN-gamma/LPS-activated RAW 264.7 cells as compared to L-NAME
Kojic acid and its manganese and zinc complexes as potential radioprotective agents
Emami S, et al.
Bioorganic & Medicinal Chemistry Letters, 17(1), 45-48 (2007)
Xiao Hu et al.
Journal of natural products, 75(1), 82-87 (2011-12-15)
Two novel 2-arylbenzofuran dimers, morusyunnansins A and B (1 and 2), two new biflavonoids, morusyunnansins C and D (3 and 4), two new flavans, morusyunnansins E and F (5 and 6), and four known flavans (7-10) were isolated from the
Wei Yi et al.
European journal of medicinal chemistry, 46(9), 4330-4335 (2011-07-23)
Melanin play a major role in human skin protection and their biosynthesis is vital. Due to their color, they contribute to the skin pigmentation. Tyrosinase is a key enzyme involved in the first stage of melanin biosynthesis, it catalyzes the
Nasz zespół naukowców ma doświadczenie we wszystkich obszarach badań, w tym w naukach przyrodniczych, materiałoznawstwie, syntezie chemicznej, chromatografii, analityce i wielu innych dziedzinach.
Skontaktuj się z zespołem ds. pomocy technicznej