Kluczowe dokumenty
E0377
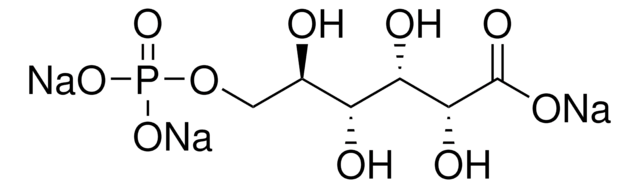
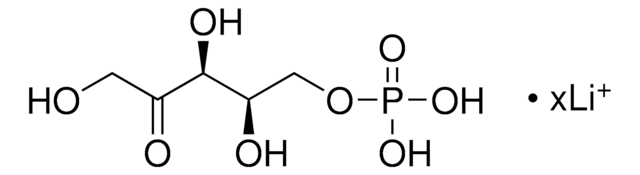
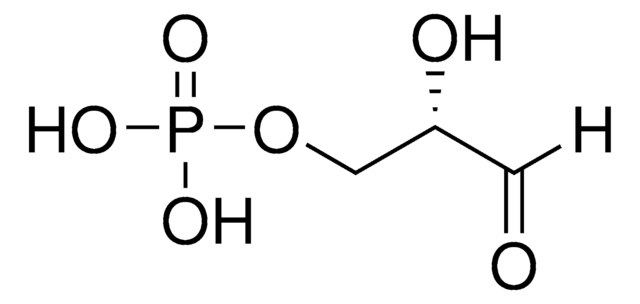
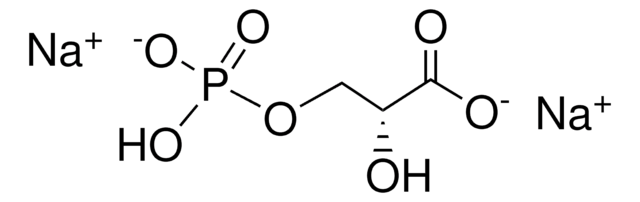
D-Erythrose 4-phosphate sodium salt
≥50% (TLC)
Synonim(y):
4-Phospho-D-erythrose sodium salt
About This Item
Polecane produkty
Poziom jakości
Próba
≥50% (TLC)
Formularz
powder
kolor
white to off-white
rozpuszczalność
water: 50 mg/mL, clear to slightly hazy, colorless to faintly yellow
temp. przechowywania
−20°C
ciąg SMILES
[Na+].[H]C(=O)[C@H](O)[C@H](O)COP(O)([O-])=O
InChI
1S/C4H9O7P.Na/c5-1-3(6)4(7)2-11-12(8,9)10;/h1,3-4,6-7H,2H2,(H2,8,9,10);/q;+1/p-1/t3-,4+;/m0./s1
Klucz InChI
KKDBADMPNGAKHM-RFKZQXLXSA-M
Szukasz podobnych produktów? Odwiedź Przewodnik dotyczący porównywania produktów
Zastosowanie
Działania biochem./fizjol.
Inne uwagi
Kod klasy składowania
11 - Combustible Solids
Klasa zagrożenia wodnego (WGK)
WGK 3
Temperatura zapłonu (°F)
Not applicable
Temperatura zapłonu (°C)
Not applicable
Wybierz jedną z najnowszych wersji:
Masz już ten produkt?
Dokumenty związane z niedawno zakupionymi produktami zostały zamieszczone w Bibliotece dokumentów.
Klienci oglądali również te produkty
Produkty
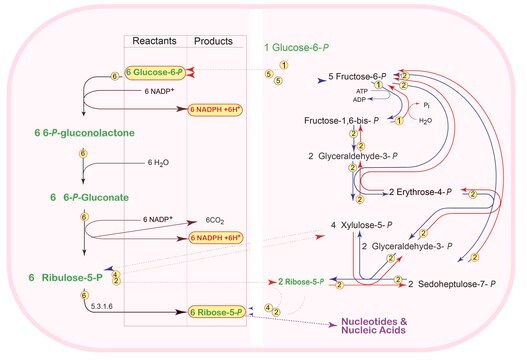
The Pentose Phosphate Pathway operates in tissues that synthesize fatty acids and steroids. Learn more about the Pentose Phosphate Pathway, what it is and how it works.
Nasz zespół naukowców ma doświadczenie we wszystkich obszarach badań, w tym w naukach przyrodniczych, materiałoznawstwie, syntezie chemicznej, chromatografii, analityce i wielu innych dziedzinach.
Skontaktuj się z zespołem ds. pomocy technicznej