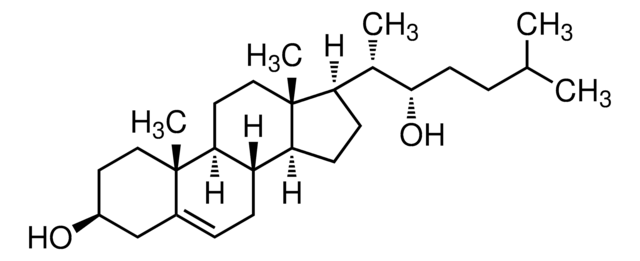
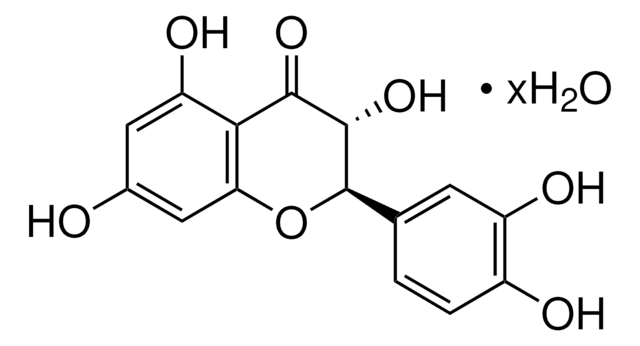
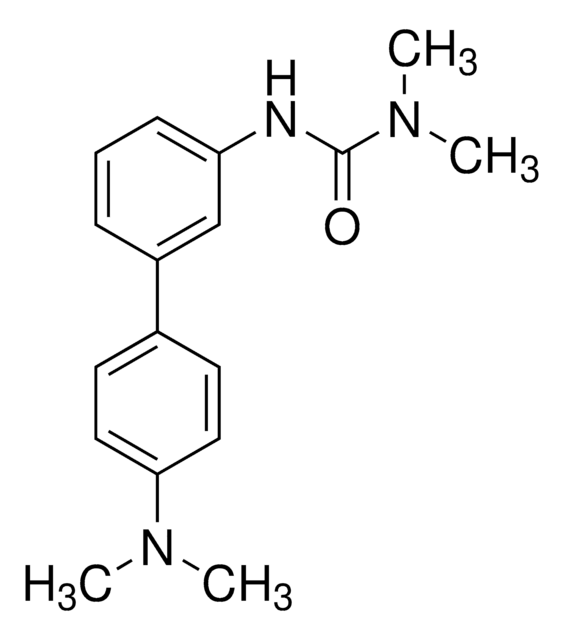
Kluczowe dokumenty
C5490
C75
≥98% (HPLC), powder
Synonim(y):
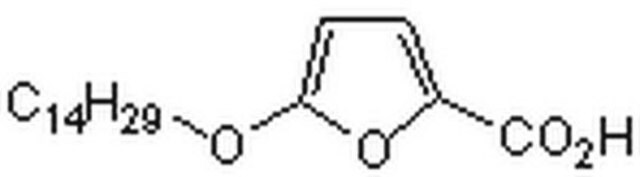
4-Methylene-2-octyl-5-oxotetrahydrofuran-3-carboxylic acid
About This Item
Polecane produkty
Próba
≥98% (HPLC)
Formularz
powder
kolor
white to beige
rozpuszczalność
DMSO: 5 mg/mL, clear
temp. przechowywania
2-8°C
ciąg SMILES
CCCCCCCCC1OC(=O)C(=C)C1C(O)=O
InChI
1S/C14H22O4/c1-3-4-5-6-7-8-9-11-12(13(15)16)10(2)14(17)18-11/h11-12H,2-9H2,1H3,(H,15,16)
Klucz InChI
VCWLZDVWHQVAJU-UHFFFAOYSA-N
Zastosowanie
- as a fatty acid synthase (FASN) inhibitor to test its ability in the direct inhibition of FASN to attenuate mammosphere formation as compared to metformin
- as a pharmacological inhibitor to inhibit fatty acid synthesis in glioma stem cells (GSCs)
- as FAS inhibitor in the pre-treatment of Chang cells to inhibit lipogenesis and reverse the senescence induced by hydrogen peroxide
Działania biochem./fizjol.
Kod klasy składowania
11 - Combustible Solids
Klasa zagrożenia wodnego (WGK)
WGK 3
Temperatura zapłonu (°F)
Not applicable
Temperatura zapłonu (°C)
Not applicable
Środki ochrony indywidualnej
Eyeshields, Gloves, type N95 (US)
Wybierz jedną z najnowszych wersji:
Masz już ten produkt?
Dokumenty związane z niedawno zakupionymi produktami zostały zamieszczone w Bibliotece dokumentów.
Klienci oglądali również te produkty
Produkty
Information on fatty acid synthesis and metabolism in cancer cells. Learn how proliferatively active cells require fatty acids for functions such as membrane generation, protein modification, and bioenergetic requirements. These fatty acids are derived either from dietary sources or are synthesized by the cell.
Nasz zespół naukowców ma doświadczenie we wszystkich obszarach badań, w tym w naukach przyrodniczych, materiałoznawstwie, syntezie chemicznej, chromatografii, analityce i wielu innych dziedzinach.
Skontaktuj się z zespołem ds. pomocy technicznej