Kluczowe dokumenty
B1758
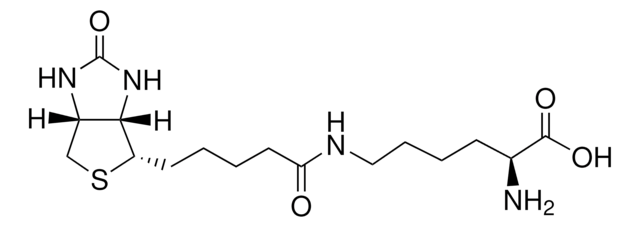
Biocytin hydrochloride
≥98% (TLC), suitable for drug transporter assays
Synonim(y):
Nε-Biotinyl-L-lysine
About This Item
Polecane produkty
Nazwa produktu
Biocytin hydrochloride, ≥98% (TLC)
Próba
≥98% (TLC)
Formularz
powder
metody
drug transporter assay: suitable
kolor
white to off-white
temp. przechowywania
−20°C
ciąg SMILES
Cl.NC(CCCCNC(=O)CCCCC1SCC2NC(=O)NC12)C(O)=O
InChI
1S/C16H28N4O4S.ClH/c17-10(15(22)23)5-3-4-8-18-13(21)7-2-1-6-12-14-11(9-25-12)19-16(24)20-14;/h10-12,14H,1-9,17H2,(H,18,21)(H,22,23)(H2,19,20,24);1H
Klucz InChI
FMGQMNFQNPUYBS-UHFFFAOYSA-N
Powiązane kategorie
Zastosowanie
- Biotynylowana amina dekstranu i chlorowodorek biocytyny są użytecznymi znacznikami do badania projekcji siatkówki u żab: Niniejszy artykuł podkreśla zastosowanie chlorowodorku biocytyny w śledzeniu projekcji siatkówki u płazów. Badanie wykazuje skuteczność chlorowodorku biocytyny w mapowaniu neuroanatomicznym, ułatwiając badania nad rozwojem i funkcją układu wzrokowego (Scalia et al., 1997).
Działania biochem./fizjol.
Kod klasy składowania
11 - Combustible Solids
Klasa zagrożenia wodnego (WGK)
WGK 3
Temperatura zapłonu (°F)
Not applicable
Temperatura zapłonu (°C)
Not applicable
Środki ochrony indywidualnej
Eyeshields, Gloves, type N95 (US)
Wybierz jedną z najnowszych wersji:
Certyfikaty analizy (CoA)
Nie widzisz odpowiedniej wersji?
Jeśli potrzebujesz konkretnej wersji, możesz wyszukać konkretny certyfikat według numeru partii lub serii.
Masz już ten produkt?
Dokumenty związane z niedawno zakupionymi produktami zostały zamieszczone w Bibliotece dokumentów.
Nasz zespół naukowców ma doświadczenie we wszystkich obszarach badań, w tym w naukach przyrodniczych, materiałoznawstwie, syntezie chemicznej, chromatografii, analityce i wielu innych dziedzinach.
Skontaktuj się z zespołem ds. pomocy technicznej

![[D-Ala2]-Leucine enkephalin ≥97% (HPLC)](/deepweb/assets/sigmaaldrich/product/structures/205/481/4fd123c0-14c7-4f90-8bdc-9e54a4bea7f1/640/4fd123c0-14c7-4f90-8bdc-9e54a4bea7f1.png)
