Kluczowe dokumenty
B4501
Biotin
≥99% (HPLC), lyophilized powder
Synonim(y):
D-Biotin, Bios II, Coenzyme R, Vitamin B7, Vitamin H
About This Item
Polecane produkty
pochodzenie biologiczne
synthetic (organic)
Poziom jakości
Próba
≥99% (HPLC)
Formularz
lyophilized powder
metody
HPLC: suitable
kolor
white to off-white
mp
231-233 °C (lit.)
rozpuszczalność
ammonium hydroxide: 50 mg/mL (2 M)
temp. przechowywania
2-8°C
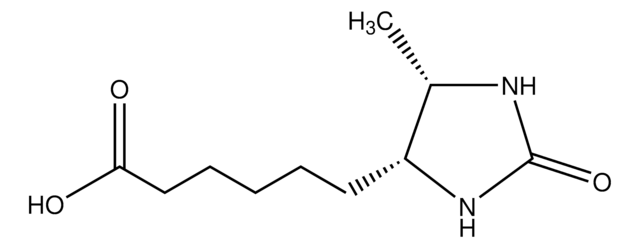
ciąg SMILES
[H][C@]12CS[C@@H](CCCCC(O)=O)[C@@]1([H])NC(=O)N2
InChI
1S/C10H16N2O3S/c13-8(14)4-2-1-3-7-9-6(5-16-7)11-10(15)12-9/h6-7,9H,1-5H2,(H,13,14)(H2,11,12,15)/t6-,7-,9-/m0/s1
Klucz InChI
YBJHBAHKTGYVGT-ZKWXMUAHSA-N
Szukasz podobnych produktów? Odwiedź Przewodnik dotyczący porównywania produktów
Opis ogólny
Zastosowanie
- for culturing of oligodendrocytes.
- as a vitamin supplement for the growth of Bacillus species.
- for blocking endogenous biotin during immunohistology procedures.
Działania biochem./fizjol.
Inne uwagi
Zastosowanie
produkt podobny
produkt powiązany
Kod klasy składowania
11 - Combustible Solids
Klasa zagrożenia wodnego (WGK)
WGK 1
Temperatura zapłonu (°F)
Not applicable
Temperatura zapłonu (°C)
Not applicable
Środki ochrony indywidualnej
Eyeshields, Gloves, type N95 (US)
Wybierz jedną z najnowszych wersji:
Certyfikaty analizy (CoA)
Nie widzisz odpowiedniej wersji?
Jeśli potrzebujesz konkretnej wersji, możesz wyszukać konkretny certyfikat według numeru partii lub serii.
Masz już ten produkt?
Dokumenty związane z niedawno zakupionymi produktami zostały zamieszczone w Bibliotece dokumentów.
Klienci oglądali również te produkty
Nasz zespół naukowców ma doświadczenie we wszystkich obszarach badań, w tym w naukach przyrodniczych, materiałoznawstwie, syntezie chemicznej, chromatografii, analityce i wielu innych dziedzinach.
Skontaktuj się z zespołem ds. pomocy technicznej