Kluczowe dokumenty
T-032
(−)-Δ8-THC solution
1.0 mg/mL in methanol, ampule of 1 mL, certified reference material, Cerilliant®
About This Item
Polecane produkty
klasa czystości
certified reference material
Poziom jakości
Formularz
liquid
Właściwości
Snap-N-Spike®/Snap-N-Shoot®
opakowanie
ampule of 1 mL
producent / nazwa handlowa
Cerilliant®
drug control
Narcotic Licence Schedule D (Switzerland); psicótropo (Spain); Decreto Lei 15/93: Tabela IIB (Portugal)
stężenie
1.0 mg/mL in methanol
metody
gas chromatography (GC): suitable
liquid chromatography (LC): suitable
Zastosowanie
cannabis testing
cannabis testing
Format
single component solution
temp. przechowywania
−20°C
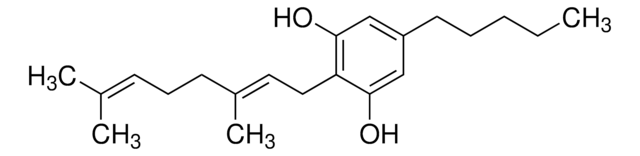
ciąg SMILES
O1C(C2C(CC(=CC2)C)c3c1cc(cc3O)CCCCC)(C)C
InChI
1S/C21H30O2/c1-5-6-7-8-15-12-18(22)20-16-11-14(2)9-10-17(16)21(3,4)23-19(20)13-15/h9,12-13,16-17,22H,5-8,10-11H2,1-4H3
Klucz InChI
HCAWPGARWVBULJ-UHFFFAOYSA-N
Opis ogólny
Zastosowanie
- Multi-analysis of 11 cannabinoids in biomass and extracts of different cannabis varieties by an HPLC-UV method, following the International Conference on Harmonization (ICH) Tripartite Guideline for Validation of Analytical Procedures
- Separation and estimation of 13 cannabinoids from different cannabis products using isocratic high-performance liquid chromatography (HPLC) in combination with a diode array detector (DAD)
- Gas chromatographic-mass spectrometric (GC-MS) analysis of ten main cannabinoids from hemp inflorescences following their silylation and esterification for derivatization
- Quantification of cannabidiol and Δ9-tetrahydrocannabinol in addition to the identification of minor cannabinoids from human plasma samples using ultra-performance liquid chromatography (UHPLC) combined with triple quadrupole mass spectrometry following a liquid-liquid extraction
- QuEChERS based extraction of 15 cannabinoids from 20 food product samples followed by their quantification by liquid chromatography-tandem mass spectrometry (LC-MS/MS)
Cechy i korzyści
- Fully characterized under ISO/IEC 17025 and ISO 17034 accreditation
- Accompanied with a comprehensive Certificate of Analysis (CoA) with data on stability, homogeneity, accuracy of concentration, uncertainty, and traceability
- Rigorously tested through real-time stability studies to ensure accuracy and shelf life
- Gravimetrically prepared using qualified precision balances to ensure minimal uncertainty
- Flame sealed under argon into ampoules for long-term shelf life
- Offered in a convenient, DEA-exempt format to improve laboratory efficiency
Informacje prawne
produkt powiązany
Hasło ostrzegawcze
Danger
Zwroty wskazujące rodzaj zagrożenia
Zwroty wskazujące środki ostrożności
Klasyfikacja zagrożeń
Acute Tox. 3 Dermal - Acute Tox. 3 Inhalation - Acute Tox. 3 Oral - Flam. Liq. 2 - STOT SE 1
Organy docelowe
Eyes
Kod klasy składowania
3 - Flammable liquids
Klasa zagrożenia wodnego (WGK)
WGK 2
Temperatura zapłonu (°F)
51.8 °F - closed cup
Temperatura zapłonu (°C)
11.0 °C - closed cup
Wykazy regulacyjne
Wykazy regulacyjne dotyczą głównie produktów chemicznych. Można w nich podawać ograniczoną liczbę informacji na temat produktów niechemicznych. Brak wpisu oznacza, że żaden ze składników nie znajduje się w wykazie. Użytkownik odpowiada za zagwarantowanie bezpiecznego i zgodnego z prawem stosowania produktu.
EU REACH Annex XVII (Restriction List)
Wybierz jedną z najnowszych wersji:
Certyfikaty analizy (CoA)
It looks like we've run into a problem, but you can still download Certificates of Analysis from our Dokumenty section.
Proszę o kontakt, jeśli potrzebna jest pomoc Obsługa Klienta
Masz już ten produkt?
Dokumenty związane z niedawno zakupionymi produktami zostały zamieszczone w Bibliotece dokumentów.
Klienci oglądali również te produkty
Produkty
As the popularity of cannabis-infused products increases, there is a growing need to characterize the type and content of the cannabinoids found in the product. This application demonstrates the ability of the Ascentis Express C18 column to baseline resolve 14 structurally-similar cannabinoids, in under seven minutes, with excellent peak shape.
CRM kannabinoidów z certyfikatem ISO, THC i THCA, dostępne jako pojedyncze składniki lub mieszanki, do dokładnego testowania mocy.
Overview of gradient HPLC-based cannabinoid analysis optimized for key method parameters.
The cannabinoids found in the Cannabis plant commonly referred to as marijuana, have grown in popularity for treating a variety of ailments from arthritis, glaucoma, and chronic pain to malnutrition, multiple sclerosis, and cancer.
Protokoły
Potency testing in marijuana-infused edibles is an important problem that analytical labs are facing due to the complexity of the involved matrices. Concentration of active ingredients in these edibles can range from a few parts per million to 3.5 parts per thousand. This application demonstrates the extraction and HPLC-UV analysis of the active compounds.
As the popularity of cannabis-infused products increases, there is a growing need to characterize the type and content of the cannabinoids found in the product. This application demonstrates the ability of the Ascentis Express C18 column to baseline resolve 14 structurally-similar cannabinoids, in under seven minutes, with excellent peak shape.
Rapid potency testing of marijuana-infused edibles using LC/MS on a biphenyl stationary phase detected eleven cannabinoids.
Tetrahydrocannabinolic acid A solution, 1.0 mg/mL in acetonitrile, ampule of 1 mL, certified reference material; Cannabichromenic Acid (CBCA) solution, 1.0 mg/mL in acetonitrile, certified reference material, ampule of 1 mL
Global Trade Item Number
| SKU | GTIN |
|---|---|
| T-032-1ML | 4061837328978 |
Nasz zespół naukowców ma doświadczenie we wszystkich obszarach badań, w tym w naukach przyrodniczych, materiałoznawstwie, syntezie chemicznej, chromatografii, analityce i wielu innych dziedzinach.
Skontaktuj się z zespołem ds. pomocy technicznej