Kluczowe dokumenty
71498
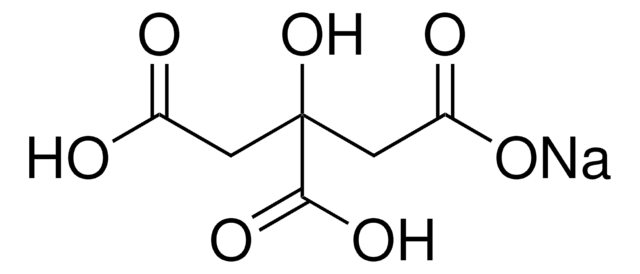
Sodium citrate monobasic
purum p.a., anhydrous, ≥99.0% (T)
Synonim(y):
Citric acid monosodium salt, Sodium dihydrogen citrate
About This Item
Polecane produkty
Poziom jakości
klasa czystości
purum p.a.
Próba
≥99.0% (T)
Formularz
crystals
pH
3.5-3.8 (25 °C, 50 mg/mL in H2O)
rozpuszczalność
water: soluble 50, clear, colorless
ślady anionów
chloride (Cl-): ≤50 mg/kg
sulfate (SO42-): ≤50 mg/kg
ślady kationów
Ca: ≤50 mg/kg
Cd: ≤50 mg/kg
Co: ≤50 mg/kg
Cu: ≤50 mg/kg
Fe: ≤50 mg/kg
K: ≤100 mg/kg
Ni: ≤50 mg/kg
Pb: ≤50 mg/kg
Zn: ≤50 mg/kg
grupa funkcyjna
carboxylic acid
hydroxyl
ciąg SMILES
[Na+].OC(=O)CC(O)(CC([O-])=O)C(O)=O
InChI
1S/C6H8O7.Na/c7-3(8)1-6(13,5(11)12)2-4(9)10;/h13H,1-2H2,(H,7,8)(H,9,10)(H,11,12);/q;+1/p-1
Klucz InChI
HWPKGOGLCKPRLZ-UHFFFAOYSA-M
Szukasz podobnych produktów? Odwiedź Przewodnik dotyczący porównywania produktów
Powiązane kategorie
Opis ogólny
Sodium citrate is a useful setting retardant for the improvement of the properties of brushite cement.
Zastosowanie
Kod klasy składowania
11 - Combustible Solids
Klasa zagrożenia wodnego (WGK)
WGK 1
Temperatura zapłonu (°F)
Not applicable
Temperatura zapłonu (°C)
Not applicable
Środki ochrony indywidualnej
Eyeshields, Gloves, type N95 (US)
Wybierz jedną z najnowszych wersji:
Masz już ten produkt?
Dokumenty związane z niedawno zakupionymi produktami zostały zamieszczone w Bibliotece dokumentów.
Klienci oglądali również te produkty
Nasz zespół naukowców ma doświadczenie we wszystkich obszarach badań, w tym w naukach przyrodniczych, materiałoznawstwie, syntezie chemicznej, chromatografii, analityce i wielu innych dziedzinach.
Skontaktuj się z zespołem ds. pomocy technicznej