Wszystkie zdjęcia(1)
Kluczowe dokumenty
About This Item
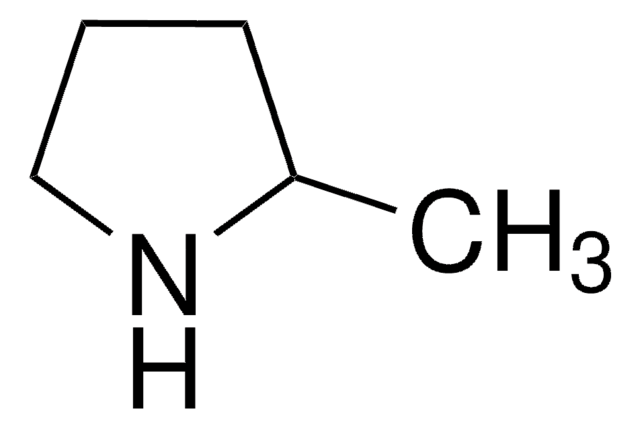
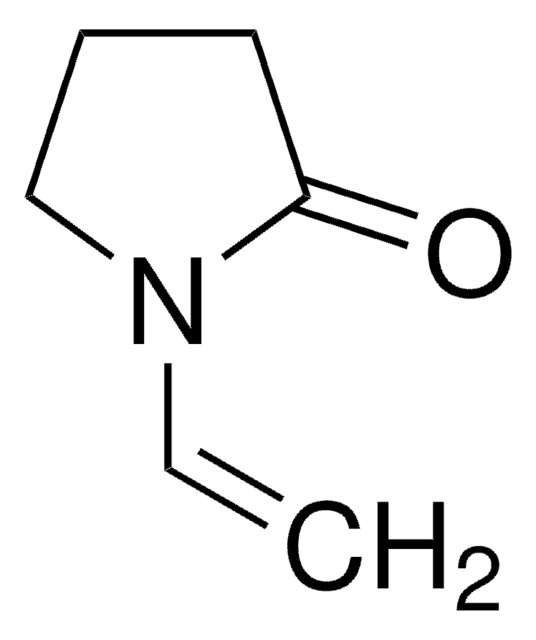
Wzór empiryczny (zapis Hilla):
C5H9N
Numer CAS:
Masa cząsteczkowa:
83.13
Numer WE:
Numer MDL:
Kod UNSPSC:
12352100
Identyfikator substancji w PubChem:
NACRES:
NA.22
Polecane produkty
Poziom jakości
Próba
95%
współczynnik refrakcji
n20/D 1.444 (lit.)
bp
104-105 °C (lit.)
gęstość
0.878 g/mL at 25 °C (lit.)
ciąg SMILES
CC1=NCCC1
InChI
1S/C5H9N/c1-5-3-2-4-6-5/h2-4H2,1H3
Klucz InChI
CTSZPNIMMLSKDV-UHFFFAOYSA-N
Powiązane kategorie
Opis ogólny
2-Methyl-1-pyrroline, a monocyclic imine, is a pyrroline derivative. It is a five-membered heterocyclic compound having various biological and pharmacological applications. It is formed during the Rh(I) complexes (containing N,N-donor ligands and N,P-donor ligand) immobilized on glassy carbon electrode surfaces catalyzed intramolecular hydroamination of 4-pentyn-1-amine. It reacts with with 2-oxopropanal to afford acetyl-1-pyrroline (AP).
Zastosowanie
2-Methyl-1-pyrroline (2-MPN) may be used in the enantioselective enzymatic synthesis of (R)- and (S)-2-methylpyrroline in the presence of whole-cells catalysts isolated from Strptomyces sp. strains GF3587 and GF3546 respectively.
Ta strona może zawierać tekst przetłumaczony maszynowo.
Hasło ostrzegawcze
Danger
Zwroty wskazujące rodzaj zagrożenia
Zwroty wskazujące środki ostrożności
Klasyfikacja zagrożeń
Eye Irrit. 2 - Flam. Liq. 2 - Skin Irrit. 2 - STOT SE 3
Organy docelowe
Respiratory system
Kod klasy składowania
3 - Flammable liquids
Klasa zagrożenia wodnego (WGK)
WGK 3
Temperatura zapłonu (°F)
51.8 °F - closed cup
Temperatura zapłonu (°C)
11 °C - closed cup
Środki ochrony indywidualnej
Eyeshields, Faceshields, Gloves, type ABEK (EN14387) respirator filter
Wybierz jedną z najnowszych wersji:
Masz już ten produkt?
Dokumenty związane z niedawno zakupionymi produktami zostały zamieszczone w Bibliotece dokumentów.
Klienci oglądali również te produkty
2-Oxopropanal, hydroxy-2-propanone, and 1-pyrroline important intermediates in the generation of the roast-smelling food flavor compounds 2-acetyl-1-pyrroline and 2-acetyltetrahydropyridine.
Hofmann T and Schieberle P.
Journal of Agricultural and Food Chemistry, 46(6), 2270-2277 (1998)
María Rodríguez-Mata et al.
Chembiochem : a European journal of chemical biology, 14(11), 1372-1379 (2013-07-03)
NADPH-dependent oxidoreductase Q1EQE0 from Streptomyces kanamyceticus catalyzes the asymmetric reduction of the prochiral monocyclic imine 2-methyl-1-pyrroline to the chiral amine (R)-2-methylpyrrolidine with >99% ee, and is thus of interest as a potential biocatalyst for the production of optically active amines.
Andrey A Tregubov et al.
Journal of the American Chemical Society, 135(44), 16429-16437 (2013-10-04)
A series of N,N-donor ligands (bis(pyrazol-1-yl)methane (bpm), bis(N-methylimidazol-2-yl)methane (bim), 1-(phenylmethyl)-4-(1H-pyrazol-1-yl methyl)-1H-1,2,3-triazole (PyT)), and one N,P-donor ligand precursor (1-(3,5-dimethylpyrazol-1-yl)(2-bromoethane) (dmPyBr)) were synthesized and functionalized with aniline. Diazotization of the aniline into an aryl diazonium, using nitrous acid in aqueous conditions, was
M Bertoldi et al.
The Biochemical journal, 342 Pt 3, 509-512 (1999-09-08)
Ornithine decarboxylase (ODC) from Lactobacillus 30a catalyses the cleavage of alpha-methylornithine into ammonia and 2-methyl-1-pyrroline; glutamate decarboxylase (GAD) from Escherichia coli catalyses the cleavage of alpha-methylglutamate into ammonia and laevulinic acid. In our analyses, 2-methyl-1-pyrroline and laevulinic acid were identified
Koichi Mitsukura et al.
Bioscience, biotechnology, and biochemistry, 75(9), 1778-1782 (2011-09-08)
The (R)-imine reductase (RIR) of Streptomyces sp. GF3587 was purified and characterized. It was found to be a NADPH-dependent enzyme, and was found to be a homodimer consisting of 32 kDa subunits. Enzymatic reduction of 10 mM 2-methyl-1-pyrroline (2-MPN) resulted
Nasz zespół naukowców ma doświadczenie we wszystkich obszarach badań, w tym w naukach przyrodniczych, materiałoznawstwie, syntezie chemicznej, chromatografii, analityce i wielu innych dziedzinach.
Skontaktuj się z zespołem ds. pomocy technicznej