すべての画像(1)
About This Item
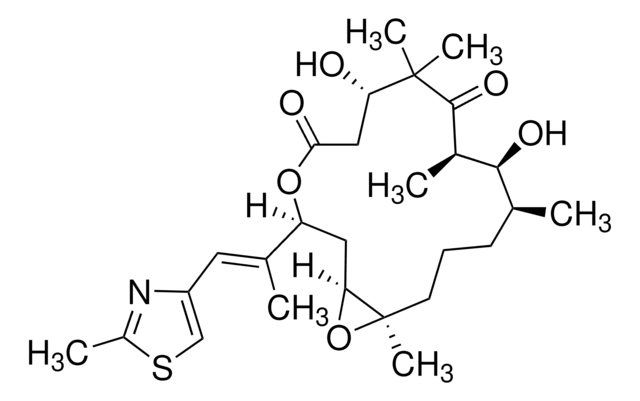
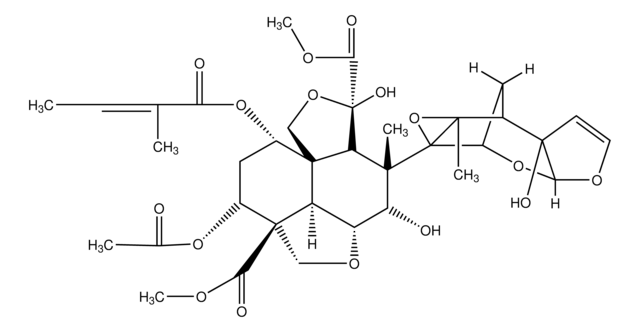
実験式(ヒル表記法):
C26H39NO6S
CAS番号:
分子量:
493.66
MDL番号:
UNSPSCコード:
12352200
PubChem Substance ID:
NACRES:
NA.77
おすすめの製品
由来生物
Sorangium cellulosum
アッセイ
>95% (HPLC)
フォーム
solid
保管条件
desiccated
protect from light
保管温度
−20°C
SMILES記法
O=C(O[C@@](/C(C)=C/C1=CSC(C)=N1)([H])C[C@@](O2)([H])[C@@]2([H])CCC[C@H](C)[C@H](O)[C@H]3C)C[C@H](O)C(C)(C)C3=O
InChI
1S/C26H39NO6S/c1-14-8-7-9-19-21(32-19)11-20(15(2)10-18-13-34-17(4)27-18)33-23(29)12-22(28)26(5,6)25(31)16(3)24(14)30/h10,13-14,16,19-22,24,28,30H,7-9,11-12H2,1-6H3/b15-10+/t14-,16+,19+,20-,21-,22-,24-/m0/s1
InChI Key
HESCAJZNRMSMJG-KKQRBIROSA-N
生物化学的/生理学的作用
(-)-Epothilone A is a microtubule (MT) stabilizing drug and natural macrolide antitumor from myxobacteria Sorangium cellulosum. EpoA exhibits kinetics similar to paclitaxel by inducing tubulin polymerization in vitro and producing enhanced microtubule stability and bundling in cultured cells. In contrast to paclitaxel, Epothilone A exhibits a greater cytotoxicity against P-glycoprotein-expressing multidrug resistant (MDR) cells (IC50 = 20 nM for MDR CCRF-CEM/VBL100 cells). Epothilone A is a competitve inhibitor of 3H-paclitaxel binding with comparable IC50 to paclitaxel in displacement competition assays. EpoA causes cell cycle arrest at the G2/M transition leading to cytotoxicity.
EpoA is a microtubule stabilizing drug and natural macrolide antitumor.
保管分類コード
11 - Combustible Solids
WGK
WGK 3
引火点(°F)
Not applicable
引火点(℃)
Not applicable
適用法令
試験研究用途を考慮した関連法令を主に挙げております。化学物質以外については、一部の情報のみ提供しています。 製品を安全かつ合法的に使用することは、使用者の義務です。最新情報により修正される場合があります。WEBの反映には時間を要することがあるため、適宜SDSをご参照ください。
Jan Code
E3656-VAR:
E3656-BULK:
E3656-10UG:
最新バージョンのいずれかを選択してください:
Daniele Passarella et al.
Bioorganic & medicinal chemistry, 17(21), 7435-7440 (2009-10-07)
The preparation and biological evaluation of a novel series of dimeric epothilone A derivatives (1-6) are described. Two types of diacyl spacers were introduced to establish the various dimeric epothilone A constructs. The effect of these compounds on tubulin polymerization
Bernhard Pfeiffer et al.
Bioorganic & medicinal chemistry letters, 19(14), 3760-3763 (2009-05-13)
The SAR of a series of new epothilone A derivatives with a 2-substituted-1,3-oxazoline moiety trans-fused to the C12-C13 bond of the deoxy macrocycle have been investigated with regard to tubulin polymerization induction and cancer cell growth inhibition. Significant differences in
Isao Kobayashi et al.
Scientific reports, 9(1), 14205-14205 (2019-10-04)
Hematopoietic stem cells (HSCs) maintain the entire blood system throughout life and are utilized in therapeutic approaches for blood diseases. Prospective isolation of highly purified HSCs is crucial to understand the molecular mechanisms underlying regulation of HSCs. The zebrafish is
Joys of molecules. 2. Endeavors in chemical biology and medicinal chemistry.
K C Nicolaou
Journal of medicinal chemistry, 48(18), 5613-5638 (2005-09-02)
Máté Erdélyi et al.
Journal of medicinal chemistry, 51(5), 1469-1473 (2008-02-15)
The conformational properties of the microtubule-stabilizing agent epothilone A ( 1a) and its 3-deoxy and 3-deoxy-2,3-didehydro derivatives 2 and 3 have been investigated in aqueous solution by a combination of NMR spectroscopic methods, Monte Carlo conformational searches, and NAMFIS calculations.
ライフサイエンス、有機合成、材料科学、クロマトグラフィー、分析など、あらゆる分野の研究に経験のあるメンバーがおります。.
製品に関するお問い合わせはこちら(テクニカルサービス)