PHR1542
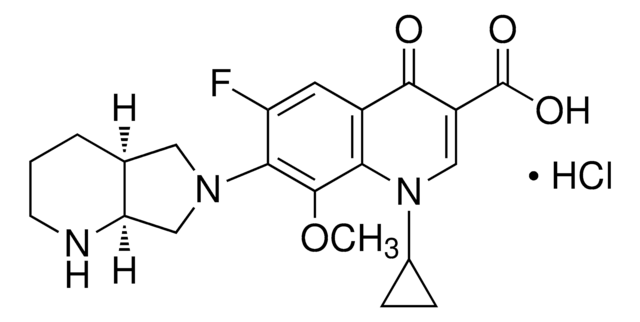
モキシフロキサシン 塩酸塩
Pharmaceutical Secondary Standard; Certified Reference Material
別名:
1-シクロプロピル-6-フルオロ-8-メトキシ-7-[(4aS,7aS)-オクタヒドロピロロ[3,4-b]ピリジン-6-イル]-4-オキソ-1,4-ジヒドロキノリン-3-カルボン酸 塩酸塩
About This Item
おすすめの製品
グレード
certified reference material
pharmaceutical secondary standard
品質水準
認証
traceable to Ph. Eur. Y0000703
traceable to USP 1448606
APIファミリー
moxifloxacin
CofA
current certificate can be downloaded
包装
pkg of 1 g
テクニック
HPLC: suitable
gas chromatography (GC): suitable
アプリケーション
pharmaceutical (small molecule)
フォーマット
neat
保管温度
2-30°C
SMILES記法
Cl.COc1c(N2C[C@@H]3CCCN[C@@H]3C2)c(F)cc4C(=O)C(=CN(C5CC5)c14)C(O)=O
InChI
1S/C21H24FN3O4.ClH/c1-29-20-17-13(19(26)14(21(27)28)9-25(17)12-4-5-12)7-15(22)18(20)24-8-11-3-2-6-23-16(11)10-24;/h7,9,11-12,16,23H,2-6,8,10H2,1H3,(H,27,28);1H/t11-,16+;/m0./s1
InChI Key
IDIIJJHBXUESQI-DFIJPDEKSA-N
類似した製品をお探しですか? 訪問 製品比較ガイド
詳細
アプリケーション
アナリシスノート
その他情報
脚注
関連製品
シグナルワード
Warning
危険有害性情報
危険有害性の分類
Acute Tox. 4 Oral - Aquatic Chronic 3 - Eye Irrit. 2
保管分類コード
11 - Combustible Solids
WGK
WGK 2
引火点(°F)
Not applicable
引火点(℃)
Not applicable
この製品を見ている人はこちらもチェック
ライフサイエンス、有機合成、材料科学、クロマトグラフィー、分析など、あらゆる分野の研究に経験のあるメンバーがおります。.
製品に関するお問い合わせはこちら(テクニカルサービス)