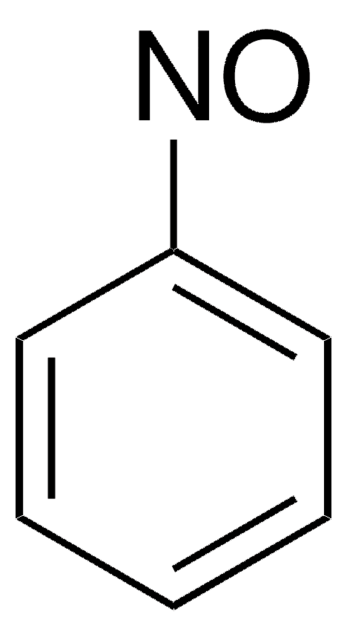
おすすめの製品
アッセイ
97%
フォーム
solid
mp
72-75 °C (lit.)
保管温度
2-8°C
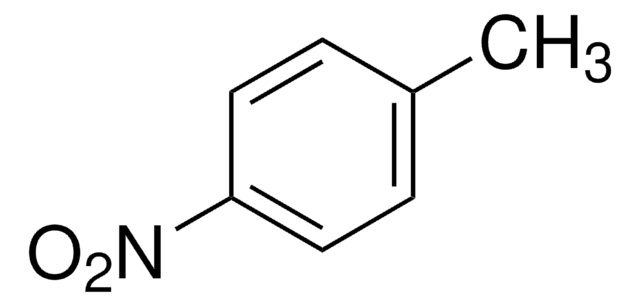
SMILES記法
Cc1ccccc1N=O
InChI
1S/C7H7NO/c1-6-4-2-3-5-7(6)8-9/h2-5H,1H3
InChI Key
TWLBRQVYXPMCFK-UHFFFAOYSA-N
関連するカテゴリー
シグナルワード
Warning
危険有害性の分類
Acute Tox. 4 Dermal - Acute Tox. 4 Inhalation - Acute Tox. 4 Oral - Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
ターゲットの組織
Respiratory system
保管分類コード
11 - Combustible Solids
WGK
WGK 3
引火点(°F)
Not applicable
引火点(℃)
Not applicable
個人用保護具 (PPE)
dust mask type N95 (US), Eyeshields, Gloves
適用法令
試験研究用途を考慮した関連法令を主に挙げております。化学物質以外については、一部の情報のみ提供しています。 製品を安全かつ合法的に使用することは、使用者の義務です。最新情報により修正される場合があります。WEBの反映には時間を要することがあるため、適宜SDSをご参照ください。
Jan Code
N26407-VAR:
N26407-25G:
N26407-5G:
N26407-BULK:
S S Hecht et al.
Cancer letters, 16(1), 103-108 (1982-05-01)
o-Toluidine hydrochloride and one of its metabolites, o-nitrosotoluene, were administered in the diet (0.028 mol/kg diet) to 2 groups of 30 male F-344 rats for 72 weeks. o-Nitrosotoluene induced significantly more tumors of the bladder (16/30 rats) and liver (20/30)
S S Hecht et al.
Cancer letters, 20(3), 349-354 (1983-10-01)
3,2'-Dimethyl-4-aminobiphenyl and 3,2'-dimethyl-4-nitrosobiphenyl were administered by subcutaneous injection in peanut oil to 2 groups of 15 male and 15 female Syrian golden hamsters. The total dose of each compound was 5.6 mmol/kg. In the group treated with 3,2'-dimethyl-4-aminobiphenyl, 24 animals
Y Ohkuma et al.
Archives of biochemistry and biophysics, 372(1), 97-106 (1999-11-24)
Mechanisms of DNA damage by metabolites of carcinogenic o-toluidine in the presence of metals were investigated by the DNA sequencing technique using (32)P-labeled human DNA fragments. 4-Amino-3-methylphenol, a major metabolite, caused DNA damage in the presence of Cu(II). Predominant cleavage
B Kulkarni et al.
Carcinogenesis, 4(10), 1275-1279 (1983-10-01)
High-performance liquid chromatography with electrochemical detection, a highly sensitive and specific method, was used to determine N-hydroxy-o-toluidine and o-toluidine in the urines of male F344 rats after the administration of 0.82 mmol/kg of o-toluidine or o-nitrosotoluene. In a six hour
Matthew A Cerny et al.
Archives of biochemistry and biophysics, 436(2), 265-275 (2005-03-31)
The inactivation of cytochrome P450 enzymes by cyclopropylamines has been attributed to a mechanism involving initial one-electron oxidation at nitrogen followed by scission of the cyclopropane ring leading to covalent modification of the enzyme. Herein, we report that in liver
ライフサイエンス、有機合成、材料科学、クロマトグラフィー、分析など、あらゆる分野の研究に経験のあるメンバーがおります。.
製品に関するお問い合わせはこちら(テクニカルサービス)