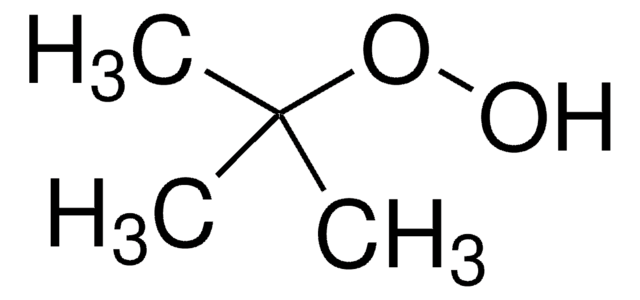
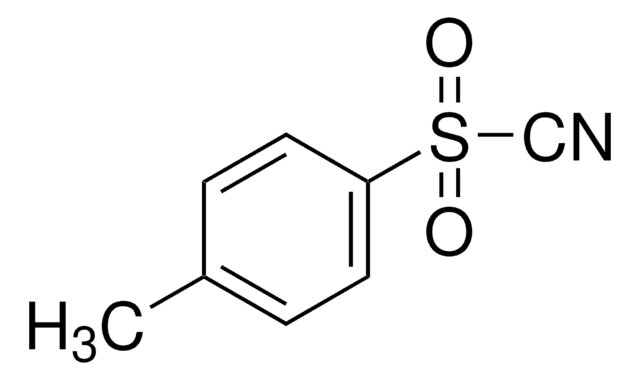
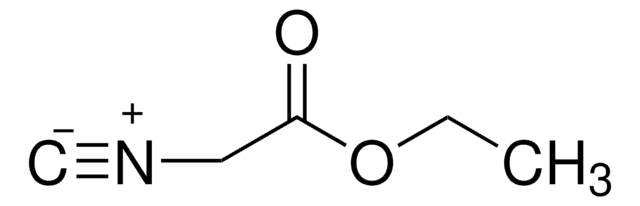
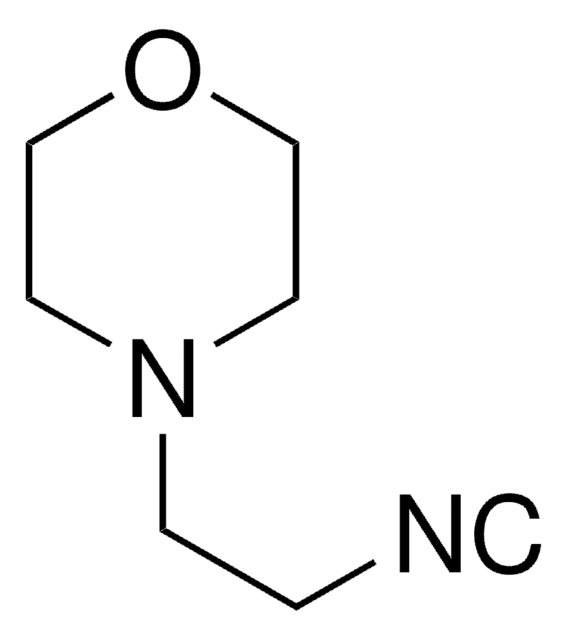
133302
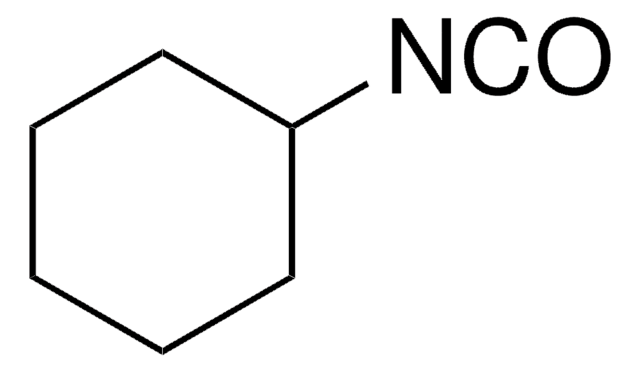
Cyclohexyl isocyanide
98%
Sinonimo/i:
Isocyanocyclohexane
About This Item
Prodotti consigliati
Livello qualitativo
Saggio
98%
Forma fisica
liquid
Indice di rifrazione
n20/D 1.45 (lit.)
Densità
0.878 g/mL at 25 °C (lit.)
Temperatura di conservazione
2-8°C
Stringa SMILE
[C-]#[N+]C1CCCCC1
InChI
1S/C7H11N/c1-8-7-5-3-2-4-6-7/h7H,2-6H2
XYZMOVWWVXBHDP-UHFFFAOYSA-N
Cerchi prodotti simili? Visita Guida al confronto tra prodotti
Descrizione generale
Applicazioni
- Isocyanide in coordination chemistry: The study on mixed "2 + 1" tricarbonyl dithiocarbamate complexes highlights Cyclohexyl isocyanide′s role as an effective monodentate ligand, contributing to advancements in radiopharmaceutical applications using Re, Tc, and Re isotopes (Shegani et al., 2021).
- Organic synthesis reagent in vascular treatments: The article discusses the use of Cyclohexyl isocyanide in modifying the endocannabinoid system, emphasizing its potential in developing treatments for conditions like hypertension through biochemical pathway modulation (Baranowska-Kuczko et al., 2021).
- Chemical process optimization in dye decolorization: Cyclohexyl isocyanide plays a crucial role in the covalent immobilization of enzymes used for the decolorization of textile dyes, demonstrating its utility in environmental chemistry and industrial applications related to pollution control (Salami et al., 2018).
- Application in nanocellulose modification: Demonstrates the versatility of Cyclohexyl isocyanide in nanotechnology by facilitating the covalent attachment of temperature-responsive polymers to cellulose nanofibrils, enhancing the material′s properties for use in smart textiles and responsive materials (Khine et al., 2018).
Avvertenze
Danger
Indicazioni di pericolo
Consigli di prudenza
Classi di pericolo
Acute Tox. 3 Dermal - Acute Tox. 3 Inhalation - Acute Tox. 3 Oral
Codice della classe di stoccaggio
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
170.6 °F - closed cup
Punto d’infiammabilità (°C)
77 °C - closed cup
Dispositivi di protezione individuale
Eyeshields, Faceshields, Gloves, type ABEK (EN14387) respirator filter
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
I clienti hanno visto anche
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.