133299
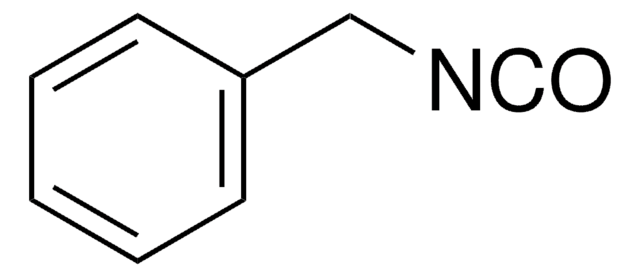
Benzyl isocyanide
98%
Sinonimo/i:
(Isocyanomethyl)benzene, Benzyl isonitrile
Autenticatiper visualizzare i prezzi riservati alla tua organizzazione & contrattuali
About This Item
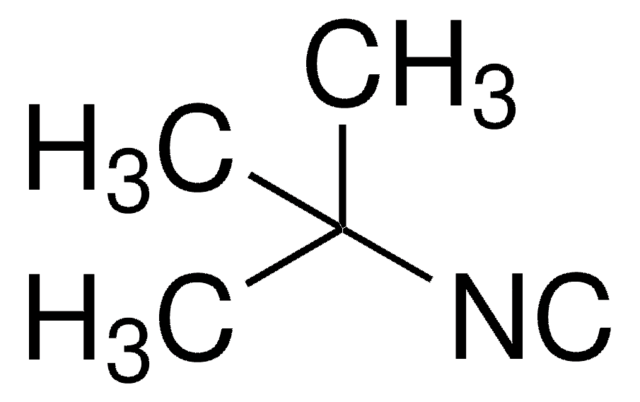
Formula condensata:
C6H5CH2NC
Numero CAS:
Peso molecolare:
117.15
Numero CE:
Numero MDL:
Codice UNSPSC:
12352100
ID PubChem:
NACRES:
NA.22
Prodotti consigliati
Livello qualitativo
Saggio
98%
Indice di rifrazione
n20/D 1.521 (lit.)
P. eboll.
105-106 °C/75 mmHg (lit.)
Densità
0.962 g/mL at 25 °C (lit.)
Gruppo funzionale
amine
isonitrile
phenyl
Temperatura di conservazione
−20°C
Stringa SMILE
[C-]#[N+]Cc1ccccc1
InChI
1S/C8H7N/c1-9-7-8-5-3-2-4-6-8/h2-6H,7H2
RIWNFZUWWRVGEU-UHFFFAOYSA-N
Categorie correlate
Descrizione generale
Benzyl isocyanide forms phosphaalkene-containing complexes.
Applicazioni
Benzyl isocyanide was used in the synthesis of Ru(II) complexes containing hydrazine and benzyl isocyanide ligands. It was used in a three-component coupling process leading to O- and N-arylamides.
Altre note
May darken in storage
Avvertenze
Warning
Indicazioni di pericolo
Consigli di prudenza
Classi di pericolo
Acute Tox. 4 Oral
Codice della classe di stoccaggio
10 - Combustible liquids
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
174.2 °F - closed cup
Punto d’infiammabilità (°C)
79 °C - closed cup
Dispositivi di protezione individuale
Eyeshields, Faceshields, Gloves, type ABEK (EN14387) respirator filter
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
I clienti hanno visto anche
Samantha N MacMillan et al.
Chemical communications (Cambridge, England), (40)(40), 4172-4174 (2007-10-11)
Reaction of (N(3)N)ZrPHPh (N(3)N=N(CH(2)CH(2)NSiMe(3))(3)(3-)) with PhCH(2)N[triple bond]C affords the 1,1-insertion product (N(3)N)Zr[C(PHPh)=NCH(2)Ph], which thermally rearranges to the phosphaalkene-containing complex, (N(3)N)Zr[N(CH(2)Ph)C(H)=PPh].
Laurent El Kaïm et al.
The Journal of organic chemistry, 72(11), 4169-4180 (2007-04-26)
The use of Smiles rearrangement in Ugi- and Passerini-type couplings with electron-deficient phenols allows very straightforward multicomponent formation of O-aryl- and N-arylamides. Best yields were observed with the highly activated o- and p-nitrophenols, salicylic derivatives giving adducts in lower yields.
Synthesis and X-ray studies of ruthenium (II) complexes containing hydrazine and benzyl isocyanide ligands.
Owalude SO, et al.
Bulletin of the Chemical Society of Ethiopia, 27(3), 405-411 (2013)
Zeinab Faghih et al.
Iranian journal of pharmaceutical research : IJPR, 19(3), 134-143 (2021-03-09)
The complex [(PhCH2NC)AuCl], 1, was prepared by the reaction of [(Me2S)AuCl], A, with an equimolar amount of benzyl isocyanide (PhCH2NC) ligand. Through a salt metathesis reaction, the chloride ligand in 1 was replaced by potassium benzothiazole-2-thiolate (Kbt) and potassium benzoimidazole-2-thiolate
Ben J Tickner et al.
Chemical science, 10(20), 5235-5245 (2019-06-14)
We report the formation of a series of novel [Ir(H)2(IMes)(α-13C2-carboxyimine)L] complexes in which the identity of the coligand L is varied. When examined with para-hydrogen, complexes in which L is benzylamine or phenethylamine show significant 1H hydride and 13C2 imine
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.