H19808
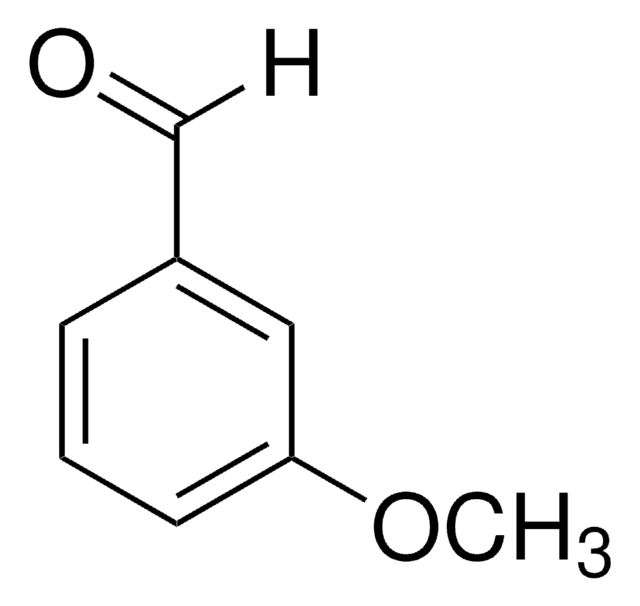
3-Hydroxybenzaldehyde
≥99%
Sinonimo/i:
3-Formylphenol, 5-Hydroxybenzaldehyde, m-Formylphenol, m-Hydroxybenzaldehyde
Autenticatiper visualizzare i prezzi riservati alla tua organizzazione & contrattuali
About This Item
Formula condensata:
HOC6H4CHO
Numero CAS:
Peso molecolare:
122.12
Beilstein:
507099
Numero CE:
Numero MDL:
Codice UNSPSC:
12352100
ID PubChem:
NACRES:
NA.22
Prodotti consigliati
Saggio
≥99%
P. ebollizione
191 °C/50 mmHg (lit.)
Punto di fusione
100-103 °C (lit.)
Stringa SMILE
Oc1cccc(C=O)c1
InChI
1S/C7H6O2/c8-5-6-2-1-3-7(9)4-6/h1-5,9H
IAVREABSGIHHMO-UHFFFAOYSA-N
Cerchi prodotti simili? Visita Guida al confronto tra prodotti
Categorie correlate
Applicazioni
3-Hydroxybenzaldehyde can be used as a reactant along with ethyl acetoacetate and thiourea in the synthesis of corresponding dihydropyrimidine-2-thione (monastrol), using Yb(OTf)3 as a catalyst by Biginelli cyclocondensation reaction.
It can also be used as a starting material for the synthesis of:
It can also be used as a starting material for the synthesis of:
- (-)-Quinocarcin, isoxazolo[3,4-e][2,1]benzisoxazole and 3-n-propylphenol.
- Oligo-3-hydroxybenzaldehyde (O-3HBA) by oxidative polycondensation.
Avvertenze
Warning
Indicazioni di pericolo
Consigli di prudenza
Classi di pericolo
Eye Irrit. 2 - Skin Irrit. 2
Codice della classe di stoccaggio
11 - Combustible Solids
Classe di pericolosità dell'acqua (WGK)
WGK 3
Dispositivi di protezione individuale
dust mask type N95 (US), Eyeshields, Gloves
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
I clienti hanno visto anche
A novel cyclization to isoxazolo [3, 4-e][2, 1] benzisoxazole
Katritzky AR, et al.
Tetrahedron Letters, 43(18), 3449-3451 (2002)
The synthesis and thermal properties of oligo-3-hydroxybenzaldehyde synthesized by oxidative polycondensation
Mart H, et al.
Journal of Applied Polymer Science, 101(2), 892-897 (2006)
A practical synthesis of 3-n-propylphenol, a component of tsetse fly attractant blends
Ujvary I and Mikite G
Organic Process Research & Development, 7(4), 585-587 (2003)
Improved synthesis and preparative scale resolution of racemic monastrol
Dondoni A, et al.
Tetrahedron Letters, 43(34), 5913-5916 (2002)
M Parekh et al.
Letters in applied microbiology, 22(2), 115-120 (1996-02-01)
Desulfovibrio desulfuricans ATCC 27774 was screened for reactivity against aromatic compounds during lactate-dependent, nitrate-dissimilating growth. Only aromatic aldehydes (benzaldehyde, 2-hydroxybenzaldehyde, 3-hydroxybenzaldehyde, 4-hydroxybenzaldehyde, vanillin, iso-vanillin and o-vanillin) were reactive and, with the exception of 2-hydroxybenzaldehyde, were stimulatory to lactate-dependent growth. Aromatic
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.