275603
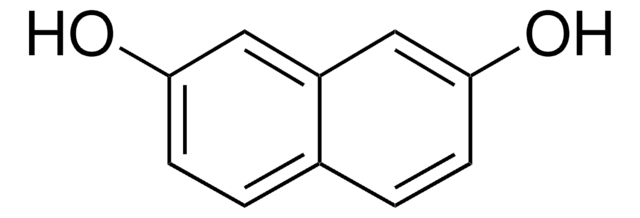
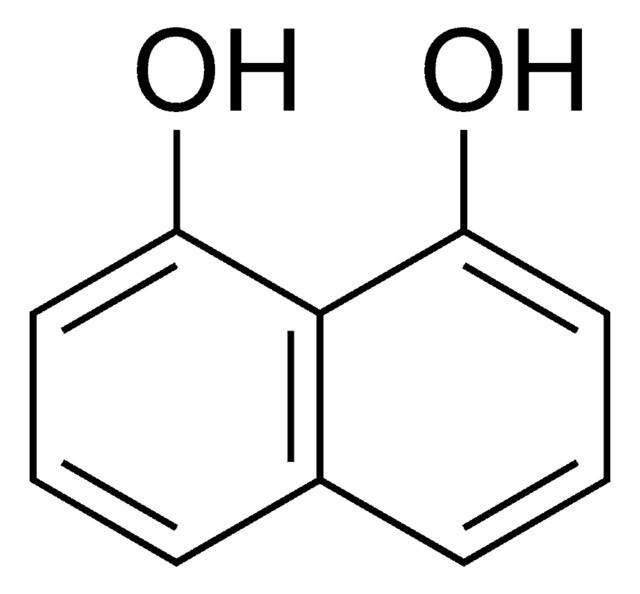
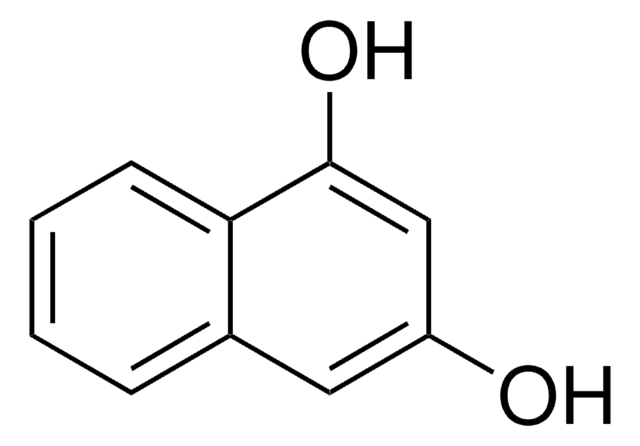
2,6-Dihydroxynaphthalene
98%
Sinonimo/i:
2,6-Naphthalenediol
Autenticatiper visualizzare i prezzi riservati alla tua organizzazione & contrattuali
About This Item
Formula condensata:
C10H6(OH)2
Numero CAS:
Peso molecolare:
160.17
Beilstein:
1238082
Numero CE:
Numero MDL:
Codice UNSPSC:
12352100
ID PubChem:
NACRES:
NA.22
Prodotti consigliati
Saggio
98%
Forma fisica
solid
Punto di fusione
223-225 °C (lit.)
Stringa SMILE
Oc1ccc2cc(O)ccc2c1
InChI
1S/C10H8O2/c11-9-3-1-7-5-10(12)4-2-8(7)6-9/h1-6,11-12H
MNZMMCVIXORAQL-UHFFFAOYSA-N
Cerchi prodotti simili? Visita Guida al confronto tra prodotti
Applicazioni
2,6-Dihydroxynaphthalene was used in the synthesis of 1,5-dichloro-2,6-diethynylnaphthalenes. It was also used in the preparation of first-generation rotaxane dendrimer.
Avvertenze
Warning
Indicazioni di pericolo
Consigli di prudenza
Classi di pericolo
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Organi bersaglio
Respiratory system
Codice della classe di stoccaggio
11 - Combustible Solids
Classe di pericolosità dell'acqua (WGK)
WGK 2
Punto d’infiammabilità (°F)
Not applicable
Punto d’infiammabilità (°C)
Not applicable
Dispositivi di protezione individuale
dust mask type N95 (US), Eyeshields, Gloves
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
I clienti hanno visto anche
Shoji Shinamura et al.
The Journal of organic chemistry, 75(4), 1228-1234 (2010-01-27)
In this paper we present the synthesis, structures, characterization, and applications to field-effect transistors (FETs) of naphtho[1,2-b:5,6-b']dithiophene (NDT) and -diselenophene (NDS) derivatives. Treatment of 1,5-dichloro-2,6-diethynylnaphthalenes, easily derived from commercially available 2,6-dihydroxynaphthalene, with sodium chalcogenide afforded a straightforward access to NDTs
Soo-Young Kim et al.
Chemistry, an Asian journal, 2(6), 747-754 (2007-05-08)
By taking advantage of the fact that cucurbit[6]uril (CB[6]) forms exceptionally stable host-guest complexes with protonated amines, and that its homologue CB[8] can encapsulate a pair of electron-rich and electron-deficient guest molecules to form a stable 1:1:1 complex, we synthesized
Alberto Macone et al.
Bioorganic & medicinal chemistry, 17(16), 6003-6007 (2009-07-21)
Aromatic substrates tyrosol (p-hydroxyphenylethanol) and 2,6-dihydroxynaphthalene (2,6-DHN) were converted into chromane derivatives by means of chemoenzymatic reactions catalyzed by the aromatic prenyltransferase of bacterial origin NovQ, using dimethylallyl bromide as allylic substrate instead of the natural isoprenyl pyrophosphate substrate. Stereoselective
Mårten Jacobsson et al.
Journal of medicinal chemistry, 49(6), 1932-1938 (2006-03-17)
The antiproliferative activity of the 14 isomeric monoxylosylated dihydroxynaphthalenes has been tested in vitro toward normal HFL-1 and 3T3 A31 cells as well as transformed T24 and 3T3 SV40 cells. The antiproliferative effect toward HFL-1 cells was correlated with the
Dieter Schemeth et al.
Analytica chimica acta, 1038, 182-190 (2018-10-04)
In this study, we focus on isolation and fractionation strategies by solid phase extraction (SPE) for a broad range of environmentally related organic acids. These emerging potential contaminants are primary degradation products of spilled petrogenic compounds but little attention has
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.