L2752
Leu-Leu
>98% (TLC), suitable for ligand binding assays
Faça loginpara ver os preços organizacionais e de contrato
About This Item
Fórmula empírica (Notação de Hill):
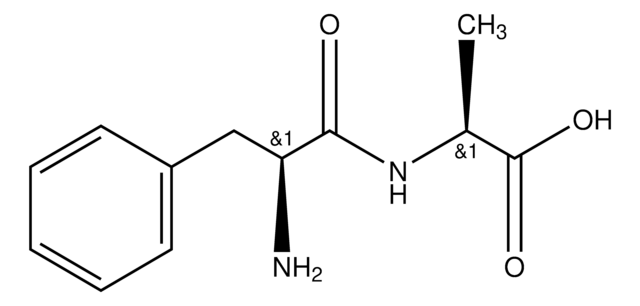
C12H24N2O3
Número CAS:
Peso molecular:
244.33
Número CE:
Número MDL:
Código UNSPSC:
12352202
ID de substância PubChem:
NACRES:
NA.26
Produtos recomendados
Nome do produto
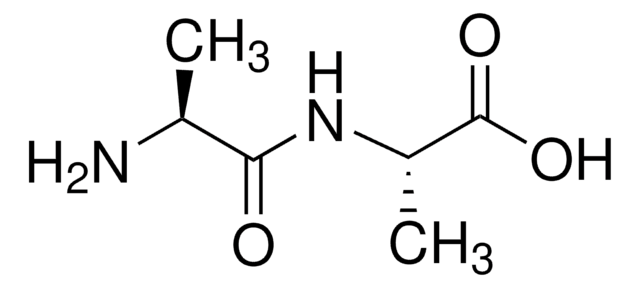
Leu-Leu,
Ensaio
>98% (TLC)
Formulário
powder
técnica(s)
ligand binding assay: suitable
cor
white to off-white
temperatura de armazenamento
−20°C
cadeia de caracteres SMILES
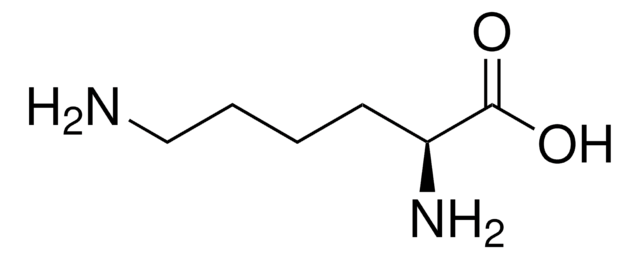
CC(C)C[C@H](N)C(=O)N[C@@H](CC(C)C)C(O)=O
InChI
1S/C12H24N2O3/c1-7(2)5-9(13)11(15)14-10(12(16)17)6-8(3)4/h7-10H,5-6,13H2,1-4H3,(H,14,15)(H,16,17)/t9-,10-/m0/s1
chave InChI
LCPYQJIKPJDLLB-UWVGGRQHSA-N
Categorias relacionadas
Amino Acid Sequence
Leu-Leu
Ações bioquímicas/fisiológicas
Leucylleucine (Leu-Leu) may be used to study the functionality of dileucine motifs such as the motif responsible for internalization and targeting of vesicular acetylcholine transporter and clathrin adaptors AP-1 and AP-2.
Código de classe de armazenamento
11 - Combustible Solids
Classe de risco de água (WGK)
WGK 3
Ponto de fulgor (°F)
Not applicable
Ponto de fulgor (°C)
Not applicable
Equipamento de proteção individual
Eyeshields, Gloves, type N95 (US)
Escolha uma das versões mais recentes:
Já possui este produto?
Encontre a documentação dos produtos que você adquiriu recentemente na biblioteca de documentos.
Lingling Zhang et al.
Oncogene, 39(49), 7196-7208 (2020-10-11)
Metastasis is responsible for the death of most breast cancer patients. Robo1 has been implicated as a tumor suppressor for various cancers including breast cancer. However, it is not well understood how Robo1 expression is regulated during tumorigenesis. In this
A van Boven et al.
Biochimie, 70(4), 535-542 (1988-04-01)
Different strains of Lactococcus lactis ssp. cremoris hydrolyze peptides at different rates while the cell-free extracts of these strains all show the same or much higher rates of hydrolysis. These observations indicate that the uptake of peptides is the rate-limiting
Yanzhen Zhou et al.
Journal of neuroinflammation, 15(1), 161-161 (2018-05-29)
Microglia-mediated neuroinflammation is recognized to mainly contribute to the pathogenesis of Parkinson's disease (PD). Tetrahydroxystilbene glucoside (TSG) has been proved to be beneficial for health with a great number of pharmacological properties. We examined the effects of TSG against dopamine
Pim W J M Frederix et al.
Dalton transactions (Cambridge, England : 2003), 41(42), 13112-13119 (2012-09-20)
A [FeFe]-hydrogenase model compound (µ-S(CH(2))(3)S)Fe(2)(CO)(4)(PMe(3))(2) [1] has been encapsulated in a low molecular weight (LMW) hydrogelator (Fmoc-Leu-Leu). Linear infrared absorption spectroscopy, gel melting and ultrafast time-resolved infrared spectroscopy experiments reveal significant contrasts in chemical environment and photochemistry between the encapsulated
Yayoi Miyashita et al.
Journal of cell science, 120(Pt 24), 4395-4406 (2007-12-07)
The E-cadherin-catenin complex regulates Ca(2+)-dependent cell-cell adhesion and is localized to the basolateral membrane of polarized epithelial cells. Uncoupling beta-catenin from E-cadherin by deletion or substitution mutations causes accumulation of these proteins in intracellular compartments, including the trans-Golgi network and
Nossa equipe de cientistas tem experiência em todas as áreas de pesquisa, incluindo Life Sciences, ciência de materiais, síntese química, cromatografia, química analítica e muitas outras.
Entre em contato com a assistência técnica