G9753
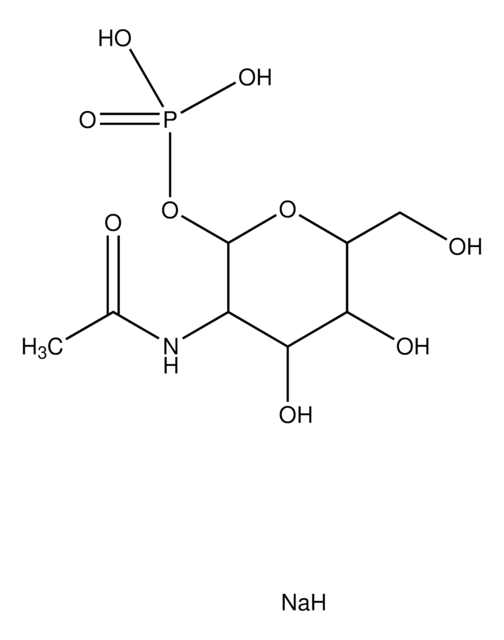
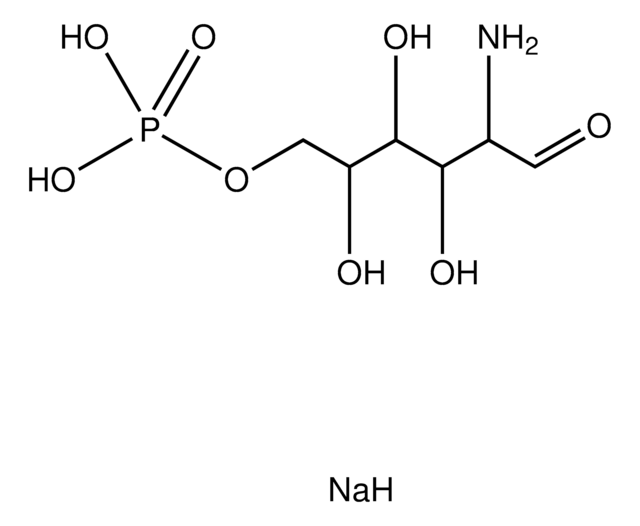
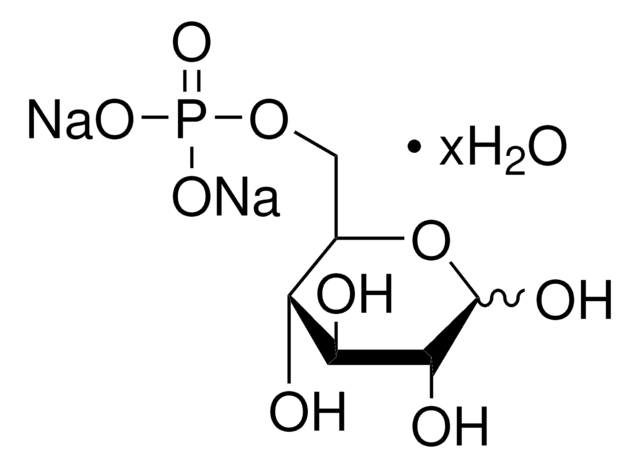
α-D-Glucosamine 1-phosphate
Sinônimo(s):
2-Amino-2-deoxy-α-D-glucopyranosyl phosphate
Faça loginpara ver os preços organizacionais e de contrato
About This Item
Fórmula empírica (Notação de Hill):
C6H14NO8P
Número CAS:
Peso molecular:
259.15
Número MDL:
Código UNSPSC:
12352201
ID de substância PubChem:
NACRES:
NA.25
Produtos recomendados
fonte biológica
natural (inorganic)
Nível de qualidade
Ensaio
≥97% (TLC)
Formulário
powder
Impurezas
<8.5% water (Karl Fischer)
cor
white
solubilidade
water: 100 mg/mL, clear, colorless
temperatura de armazenamento
−20°C
cadeia de caracteres SMILES
NC1C(O)C(O)C(CO)OC1OP(O)(O)=O
InChI
1S/C6H14NO8P/c7-3-5(10)4(9)2(1-8)14-6(3)15-16(11,12)13/h2-6,8-10H,1,7H2,(H2,11,12,13)
chave InChI
YMJBYRVFGYXULK-UHFFFAOYSA-N
Aplicação
- Cellodextrin phosphorylase from Ruminiclostridium thermocellum: X-ray crystal structure and substrate specificity analysis. This study presents the enzymatic synthesis and analysis of alpha-ᴅ-Glucosamine 1-phosphate based polysaccharides using cellodextrin phosphorylase, showcasing potential for novel biomaterial development. Field et al., 2017
- Glucose-1-phosphate uridylyltransferase from Erwinia amylovora: Activity, structure and substrate specificity. This paper explores the biochemical pathway involving alpha-ᴅ-Glucosamine 1-phosphate in the context of bacterial metabolism, providing insights into microbial biochemistry and potential targets for antibacterial therapy. Field et al., 2017
Outras notas
To gain a comprehensive understanding of our extensive range of Monosaccharides for your research, we encourage you to visit our Carbohydrates Category page.
Código de classe de armazenamento
11 - Combustible Solids
Classe de risco de água (WGK)
WGK 3
Ponto de fulgor (°F)
Not applicable
Ponto de fulgor (°C)
Not applicable
Equipamento de proteção individual
Eyeshields, Gloves, type N95 (US)
Escolha uma das versões mais recentes:
Já possui este produto?
Encontre a documentação dos produtos que você adquiriu recentemente na biblioteca de documentos.
Os clientes também visualizaram
D Mengin-Lecreulx et al.
The Journal of biological chemistry, 271(1), 32-39 (1996-01-05)
Two different approaches to identify the gene encoding the phosphoglucosamine mutase in Escherichia coli were used: (i) the purification to near homogeneity of this enzyme from a wild type strain and the determination of its N-terminal amino acid sequence; (ii)
Fumitaka Kudo et al.
Journal of the American Chemical Society, 127(6), 1711-1718 (2005-02-11)
Aminoglycoside antibiotics are composed of aminosugars and a unique aminocyclitol aglycon including 2-deoxystreptamine (DOS), streptidine, actinamine, etc., and nucleotidylyltransferases, sugar modifying enzymes, and glycosyltransferases appear to be essential for their biosynthesis. However, the genes encoding those enzymes were unable to
E V Vorob'eva et al.
Bioorganicheskaia khimiia, 32(5), 538-545 (2006-10-18)
The hydrolysis of defatted cells of the marine bacterium Chryseobacterium scophtalmum CIP 104199T with 10% acetic acid (3 h, 100 degrees C) led to an unusual lipid A (LA) (yield 0.6%), obtained for the first time. Using chemical analysis, FAB
S Ambrosio et al.
Journal of biochemical and biophysical methods, 25(4), 237-244 (1992-12-01)
Galactosamine is quickly metabolized to galactosamine 1-phosphate in rats treated with this compound. An HPLC method to quantify hexosamine phosphates in biological samples is described, modified from the o-phthaldialdehyde amino acid analysis procedure. o-Phthaldialdehyde derivatives of hexosamines and hexosamine-phosphates can
Seema C Namboori et al.
Journal of bacteriology, 190(8), 2987-2996 (2008-02-12)
Archaea and eukaryotes share a dolichol phosphate-dependent system for protein N-glycosylation. In both domains, the acetamido sugar N-acetylglucosamine (GlcNAc) forms part of the core oligosaccharide. However, the archaeal Methanococcales produce GlcNAc using the bacterial biosynthetic pathway. Key enzymes in this
Nossa equipe de cientistas tem experiência em todas as áreas de pesquisa, incluindo Life Sciences, ciência de materiais, síntese química, cromatografia, química analítica e muitas outras.
Entre em contato com a assistência técnica