W359602
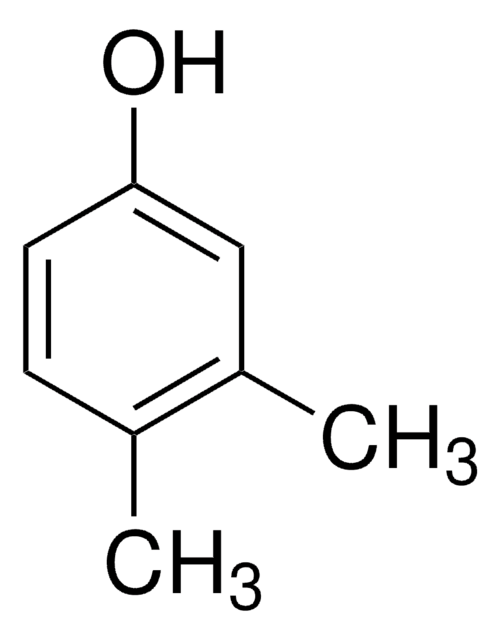
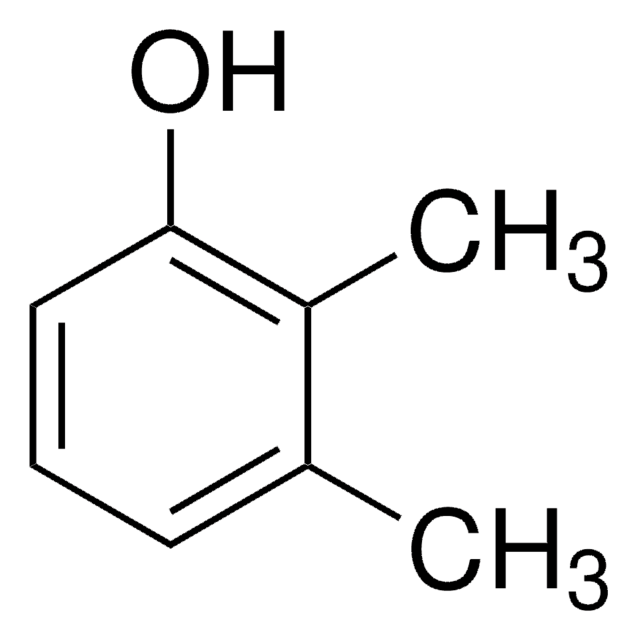
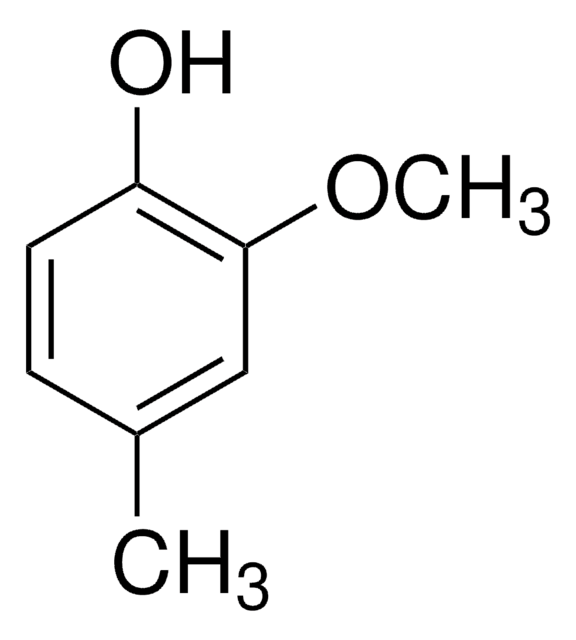
3,4-Xylenol
≥98%, FG
Sinônimo(s):
3,4-Dimethylphenol, 4-Hydroxy-o-xylene
About This Item
Produtos recomendados
fonte biológica
synthetic
Nível de qualidade
grau
FG
Fragrance grade
Halal
Agency
follows IFRA guidelines
meets purity specifications of JECFA
conformidade reg.
EU Regulation 1223/2009
EU Regulation 1334/2008 & 872/2012
Ensaio
≥98%
p.e.
227 °C (lit.)
aplicação(ões)
flavors and fragrances
Documentação
see Safety & Documentation for available documents
alérgeno alimentar
no known allergens
alérgeno de fragrância
no known allergens
Organoléptico
burnt
cadeia de caracteres SMILES
Cc1ccc(O)cc1C
InChI
1S/C8H10O/c1-6-3-4-8(9)5-7(6)2/h3-5,9H,1-2H3
chave InChI
YCOXTKKNXUZSKD-UHFFFAOYSA-N
Procurando produtos similares? Visita Guia de comparação de produtos
Categorias relacionadas
Aplicação
- Green Aromatic Epoxidation with an Iron Porphyrin Catalyst for One-Pot Functionalization of Renewable Xylene, Quinoline, and Acridine.: This study explores the green epoxidation of aromatic compounds using an iron porphyrin catalyst, demonstrating effective one-pot functionalization of renewable xylene derivatives, including 3,4-xylenol (Corrêa et al., 2023).
- A highly expressed odorant receptor from the yellow fever mosquito, AaegOR11, responds to (+)- and (-)-fenchone and a phenolic repellent.: This research identifies an odorant receptor in mosquitoes that responds to various compounds, including 3,4-dimethylphenol, which could have implications for developing new repellents (Lu et al., 2022).
Palavra indicadora
Danger
Frases de perigo
Declarações de precaução
Classificações de perigo
Acute Tox. 3 Dermal - Acute Tox. 3 Oral - Aquatic Chronic 2 - Eye Dam. 1 - Skin Corr. 1B
Código de classe de armazenamento
6.1A - Combustible acute toxic Cat. 1 and 2 / very toxic hazardous materials
Classe de risco de água (WGK)
WGK 3
Ponto de fulgor (°F)
230.0 °F - closed cup
Ponto de fulgor (°C)
110 °C - closed cup
Equipamento de proteção individual
Eyeshields, Faceshields, Gloves, type P3 (EN 143) respirator cartridges
Escolha uma das versões mais recentes:
Já possui este produto?
Encontre a documentação dos produtos que você adquiriu recentemente na biblioteca de documentos.
Os clientes também visualizaram
Global Trade Item Number
| SKU | GTIN |
|---|---|
| W359602-1KG | 4061835567201 |
| W359602-250G | 4061835567218 |
| W359602-5KG | 4061835567225 |
| W359602-SAMPLE | 4061837537004 |
Nossa equipe de cientistas tem experiência em todas as áreas de pesquisa, incluindo Life Sciences, ciência de materiais, síntese química, cromatografia, química analítica e muitas outras.
Entre em contato com a assistência técnica