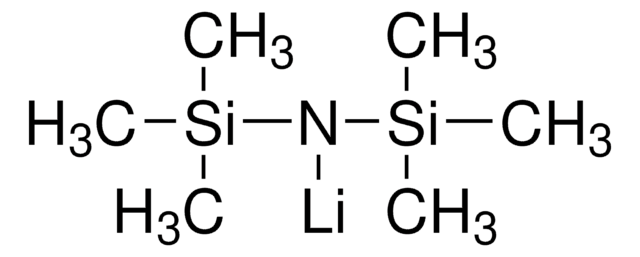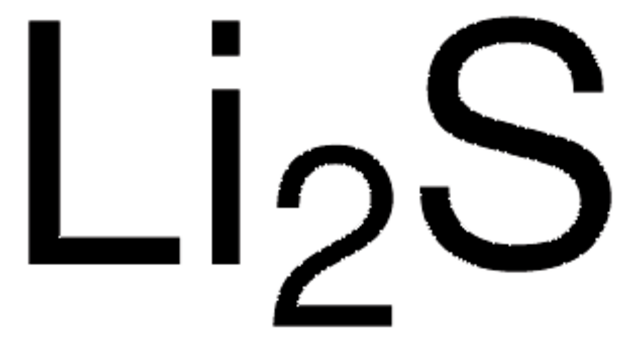930938
Lithium nitrate
battery grade, 99.999% trace metals basis
Sinônimo(s):
Lithium salt of nitric acid
About This Item
Produtos recomendados
grau
battery grade
Nível de qualidade
Ensaio
99.999% trace metals basis
Formulário
powder
características do produto alternativo mais ecológico
Design for Energy Efficiency
Learn more about the Principles of Green Chemistry.
sustainability
Greener Alternative Product
Impurezas
≤15 ppm (trace metals analysis)
pf
264 °C (lit.)
solubilidade
soluble (H2O: highly soluble(lit.); alcohols: soluble(lit.); acetone: soluble(lit.))
aplicação(ões)
battery manufacturing
cadeia de caracteres SMILES
[Li+].[O-][N+]([O-])=O
InChI
1S/Li.NO3/c;2-1(3)4/q+1;-1
chave InChI
IIPYXGDZVMZOAP-UHFFFAOYSA-N
Procurando produtos similares? Visita Guia de comparação de produtos
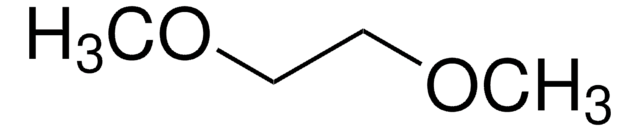
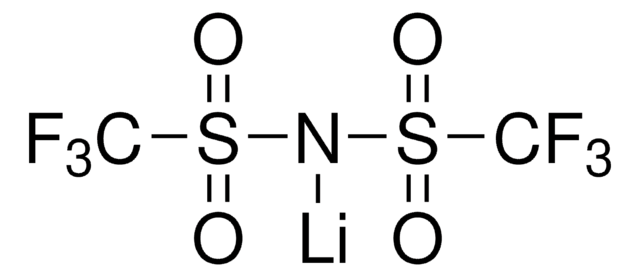
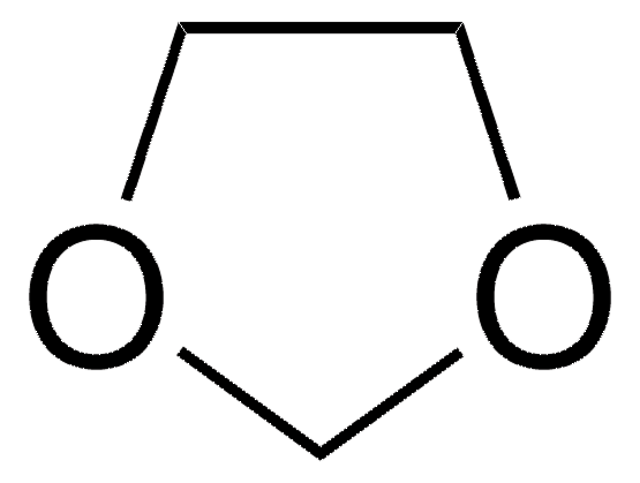
Categorias relacionadas
Descrição geral
Lithium nitrate is produced by reacting nitric acid and lithium carbonate, which evolves carbon dioxide and water. The resulting material is purified and dried.
Aplicação
Our 99.999% lithium nitrate is also well-suited for use as an additive to electrolytes in lithium-sulfur batteries and lithium metal batteries. Lithium nitrate can passivate the surface of lithium metal and suppress the redox shuttle of the dissolved lithium polysulfides on the lithium anode. In one study, the addition of 0.3 M LiNO3 nearly doubled the gravimetric capacity of lithium-sulfide batteries. Another study found that the dissolution of 1 to 5 wt% LiNO3 to the electrolyte suppressed growth of lithium dendrites and extended cycle lifetimes. Similarly beneficial effects of lithium nitrate as an additive have been observed with Li2S cathodes, carbon nanofiber-encapsulated sulfur cathodes, cobalt sulfide (Co3S4) cathodes, and polyacrylonitrile-sulfur composite cathodes. Even lithium metal anodes with LiNi0.8Co0.15Al0.05O2 (NCA) cathodes with LiNO3 added to the electrolyte showed higher coulombic efficiencies and suppressed dendrite formation compared to the electrolyte without LiNO3.
Palavra indicadora
Warning
Frases de perigo
Declarações de precaução
Classificações de perigo
Acute Tox. 4 Oral - Eye Irrit. 2 - Ox. Sol. 3
Código de classe de armazenamento
5.1B - Oxidizing hazardous materials
Classe de risco de água (WGK)
WGK 1
Ponto de fulgor (°F)
Not applicable
Ponto de fulgor (°C)
Not applicable
Escolha uma das versões mais recentes:
Certificados de análise (COA)
It looks like we've run into a problem, but you can still download Certificates of Analysis from our Documentos section.
Se precisar de ajuda, entre em contato Atendimento ao cliente
Já possui este produto?
Encontre a documentação dos produtos que você adquiriu recentemente na biblioteca de documentos.
Global Trade Item Number
| SKU | GTIN |
|---|---|
| 930938-10G | 4065268021101 |
| 930938-50G | 4065268021118 |
Nossa equipe de cientistas tem experiência em todas as áreas de pesquisa, incluindo Life Sciences, ciência de materiais, síntese química, cromatografia, química analítica e muitas outras.
Entre em contato com a assistência técnica