489174
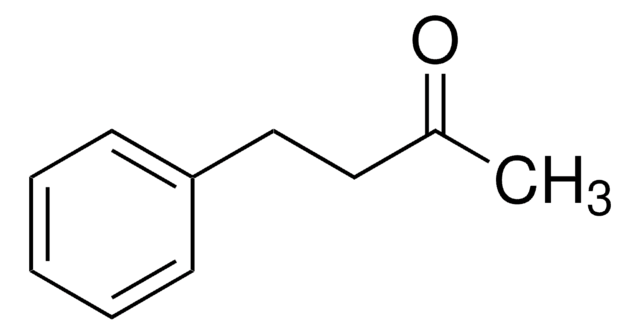
Benzyloxyacetone
90%
Sinônimo(s):
1-Benzyloxy-2-propanone
Faça loginpara ver os preços organizacionais e de contrato
About This Item
Fórmula linear:
C6H5CH2OCH2COCH3
Número CAS:
Peso molecular:
164.20
Número MDL:
Código UNSPSC:
12352100
ID de substância PubChem:
NACRES:
NA.22
Produtos recomendados
Ensaio
90%
Impurezas
3% (benzyloxy)acetic acid
3% benzyl alcohol
índice de refração
n20/D 1.512 (lit.)
p.e.
259 °C (lit.)
densidade
1.04 g/mL at 25 °C (lit.)
grupo funcional
ether
ketone
phenyl
cadeia de caracteres SMILES
CC(=O)COCc1ccccc1
InChI
1S/C10H12O2/c1-9(11)7-12-8-10-5-3-2-4-6-10/h2-6H,7-8H2,1H3
chave InChI
YHMRKVGUSQWDGZ-UHFFFAOYSA-N
Categorias relacionadas
Descrição geral
Benzyloxyacetone (α-Benzyloxyacetone) is an α-substituted acetone. It undergoes direct aldol reaction with 4-nitrobenzaldehyde in the presence of (S)-BINAM-L-prolinamide/benzoic acid to form predominantly the syn-diasteroisomer.
Aplicação
Benzyloxyacetone (1-Benzyloxy-2-propanone) may be used in the synthesis of:
- 7-benzyloxy-6-methyl-5-hepten-1-yne
- (Z)-2-methylhept-2-en-6-yn-1-o1
- (S)-(+)-1,2-propanediol, 1-benzyl ether
Código de classe de armazenamento
10 - Combustible liquids
Classe de risco de água (WGK)
WGK 3
Ponto de fulgor (°F)
230.0 °F - closed cup
Ponto de fulgor (°C)
110 °C - closed cup
Equipamento de proteção individual
Eyeshields, Gloves, multi-purpose combination respirator cartridge (US)
Escolha uma das versões mais recentes:
Já possui este produto?
Encontre a documentação dos produtos que você adquiriu recentemente na biblioteca de documentos.
Synthesis and biological evaluation of L-and D-configurations of 2',3'-dideoxy-4'-C-methyl-3'-oxacytidine analogues.
Liu MC, et al.
Bioorganic & Medicinal Chemistry Letters, 11(17), 2301-2304 (2001)
A stereoselective formation of (Z)-2-methyl-2-alkenol by the wittig reaction: its application to a synthesis of nerylacetone and (Z,Z)-farnesylacetone.
Sato K, et al.
Chemistry Letters (Jpn), 10(12), 1711-1714 (1981)
Cis selective wittig olefination of a-alkoxy ketones and its application to the stereoselective synthesis of plaunotol.
Inoue S, et al.
Bulletin of the Chemical Society of Japan, 63(6), 1629-1635 (1990)
Highly selective direct aldol reaction organocatalyzed by (S)-BINAM-L-prolinamide and benzoic acid using a-chalcogen-substituted ketones as donors.
Guillena G, et al.
ARKIVOC (Gainesville, FL, United States), 4, 260-269 (2007)
Stereochemical control of bakers' yeast mediated reduction of a protected 2-hydroxy ketone.
Manzocchi A, et al.
The Journal of Organic Chemistry, 53(18), 4405-4407 (1988)
Nossa equipe de cientistas tem experiência em todas as áreas de pesquisa, incluindo Life Sciences, ciência de materiais, síntese química, cromatografia, química analítica e muitas outras.
Entre em contato com a assistência técnica