461350
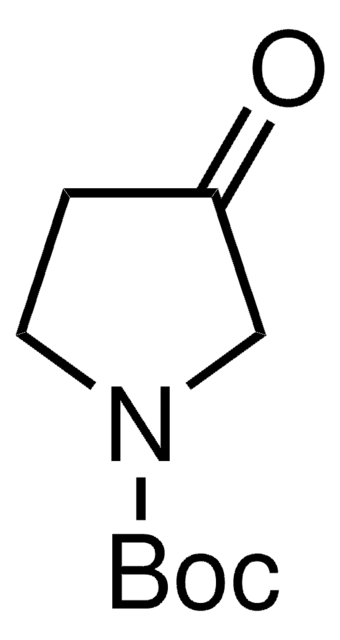
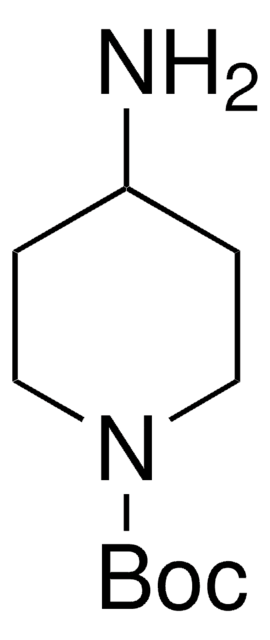
1-Boc-4-piperidone
98%
Sinônimo(s):
N-Boc-4-piperidone, tert-Butyl 4-oxo-1-piperidinecarboxylate
Faça loginpara ver os preços organizacionais e de contrato
About This Item
Fórmula empírica (Notação de Hill):
C10H17NO3
Número CAS:
Peso molecular:
199.25
Beilstein:
3650236
Número MDL:
Código UNSPSC:
12352100
ID de substância PubChem:
NACRES:
NA.22
Produtos recomendados
Nível de qualidade
Ensaio
98%
pf
73-77 °C (lit.)
grupo funcional
ketone
cadeia de caracteres SMILES
CC(C)(C)OC(=O)N1CCC(=O)CC1
InChI
1S/C10H17NO3/c1-10(2,3)14-9(13)11-6-4-8(12)5-7-11/h4-7H2,1-3H3
chave InChI
ROUYFJUVMYHXFJ-UHFFFAOYSA-N
Procurando produtos similares? Visita Guia de comparação de produtos
Descrição geral
1-Boc-4-piperidone is used as a precursor in the synthesis of synthetic drugs and as a building block for the preparation of selective ligands.
Aplicação
1-Boc-4-piperidone, a pharma building block, may be used in the synthesis of (3E,5E)-3,5-bis(2,5-dimethoxybenzylidene)-1-t-butoxycarbonylpiperidin-4-one (RL197). It may also be used in the synthesis of spirorifamycins containing a piperidine ring structure.
Advertência
Avoid metal contact.
Código de classe de armazenamento
11 - Combustible Solids
Classe de risco de água (WGK)
WGK 3
Ponto de fulgor (°F)
Not applicable
Ponto de fulgor (°C)
Not applicable
Equipamento de proteção individual
Eyeshields, Gloves, type N95 (US)
Escolha uma das versões mais recentes:
Já possui este produto?
Encontre a documentação dos produtos que você adquiriu recentemente na biblioteca de documentos.
In Ho Kim et al.
Bioorganic & medicinal chemistry letters, 17(5), 1181-1184 (2006-12-27)
A novel series of spirorifamycins was synthesized and their antibacterial activity evaluated both in vitro and in vivo. This new series of rifamycins shows excellent activity against Staphylococcus aureus that is equivalent to rifabutin. However, some compounds of the series
Atsuya Takami et al.
Bioorganic & medicinal chemistry, 12(9), 2115-2137 (2004-04-15)
Several structurally unrelated scaffolds of the Rho kinase inhibitor were designed using pharmacophore information obtained from the results of a high-throughput screening and structural information from a homology model of Rho kinase. A docking simulation using the ligand-binding pocket of
John W Clader et al.
Bioorganic & medicinal chemistry, 12(2), 319-326 (2004-01-16)
Anthranilamide analogues such as 23 are potent and highly selective muscarinic M2 antagonists that also show good oral bioavailability and in vivo activity.
Marlon Cowart et al.
Journal of medicinal chemistry, 47(15), 3853-3864 (2004-07-09)
A new class of agents with potential utility for the treatment of erectile dysfunction has been discovered, guided by the hypothesis that selective D4 agonists are erectogenic but devoid of the side effects typically associated with dopaminergic agents. The lead
Gebhard Thoma et al.
Journal of medicinal chemistry, 47(8), 1939-1955 (2004-04-02)
The chemokine receptor CCR5 plays an important role in inflammatory and autoimmune disorders as well as in transplant rejection by affecting the trafficking of effector T cells and monocytes to diseased tissues. Antagonists of CCR5 are believed to be of
Nossa equipe de cientistas tem experiência em todas as áreas de pesquisa, incluindo Life Sciences, ciência de materiais, síntese química, cromatografia, química analítica e muitas outras.
Entre em contato com a assistência técnica