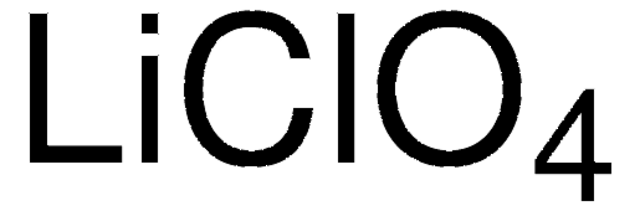431567
Lithium perchlorate
99.99% trace metals basis
Sinônimo(s):
Perchloric acid, lithium salt
About This Item
Produtos recomendados
grau
ACS reagent
Nível de qualidade
Ensaio
99.99% trace metals basis
Formulário
granular
adequação da reação
reagent type: oxidant
características do produto alternativo mais ecológico
Design for Energy Efficiency
Learn more about the Principles of Green Chemistry.
sustainability
Greener Alternative Product
Impurezas
≤0.005% insolubles
<100 ppm total metallic impurities
pH
6.0-7.5
pf
236 °C (lit.)
solubilidade
H2O: 106.4 g/L at 20 °C
traços de ânion
chloride (Cl-): ≤0.003%
sulfate (SO42-): ≤0.001%
categoria alternativa mais ecológica
cadeia de caracteres SMILES
[Li+].[O-]Cl(=O)(=O)=O
InChI
1S/ClHO4.Li/c2-1(3,4)5;/h(H,2,3,4,5);/q;+1/p-1
chave InChI
MHCFAGZWMAWTNR-UHFFFAOYSA-M
Procurando produtos similares? Visita Guia de comparação de produtos
Categorias relacionadas
Descrição geral
Aplicação
- As a precursor to prepare solid polymer electrolytes for rechargeable Li-ion batteries.
- As an oxidizer to prepare polymer-based solid propellants.
- To fabricate cobalt sulfide (CoS)-based counter electrode for dye-sensitized solar cells(DSSC).
Palavra indicadora
Danger
Frases de perigo
Declarações de precaução
Classificações de perigo
Acute Tox. 4 Oral - Eye Dam. 1 - Ox. Sol. 2 - Skin Corr. 1A - STOT SE 3
Órgãos-alvo
Respiratory system
Código de classe de armazenamento
5.1A - Strongly oxidizing hazardous materials
Classe de risco de água (WGK)
WGK 1
Ponto de fulgor (°F)
Not applicable
Ponto de fulgor (°C)
Not applicable
Equipamento de proteção individual
Eyeshields, Gloves, type P3 (EN 143) respirator cartridges
Escolha uma das versões mais recentes:
Já possui este produto?
Encontre a documentação dos produtos que você adquiriu recentemente na biblioteca de documentos.
Os clientes também visualizaram
Nossa equipe de cientistas tem experiência em todas as áreas de pesquisa, incluindo Life Sciences, ciência de materiais, síntese química, cromatografia, química analítica e muitas outras.
Entre em contato com a assistência técnica