274852
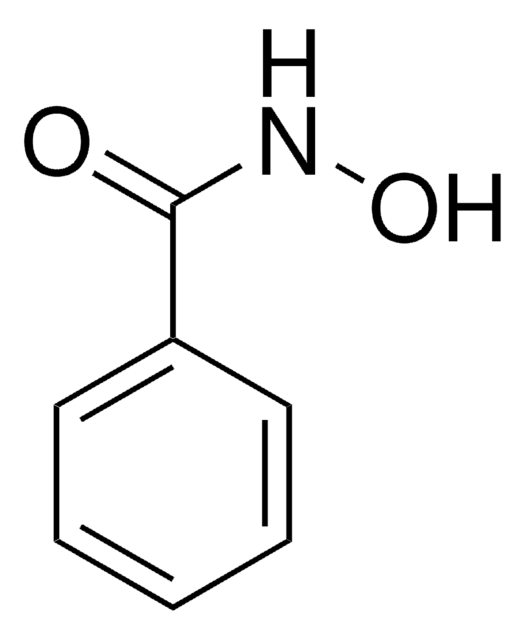
N-Benzoyl-N-phenylhydroxylamine
98%
Sinônimo(s):
N-Hydroxy-N-phenylbenzamide, N-Phenylbenzohydroxamic acid
Faça loginpara ver os preços organizacionais e de contrato
About This Item
Fórmula linear:
C6H5CON(OH)C6H5
Número CAS:
Peso molecular:
213.23
Beilstein:
2212449
Número CE:
Número MDL:
Código UNSPSC:
12352100
ID de substância PubChem:
NACRES:
NA.22
Produtos recomendados
Nível de qualidade
Ensaio
98%
Formulário
powder
pf
118-120 °C (lit.)
temperatura de armazenamento
2-8°C
cadeia de caracteres SMILES
ON(c1ccccc1)C(=O)c2ccccc2
InChI
1S/C13H11NO2/c15-13(11-7-3-1-4-8-11)14(16)12-9-5-2-6-10-12/h1-10,16H
chave InChI
YLYIXDZITBMCIW-UHFFFAOYSA-N
Procurando produtos similares? Visita Guia de comparação de produtos
Categorias relacionadas
Descrição geral
The mechanism of the reaction of N-benzoyl-N-phenylhydroxylamine with vanadium(IV) was studied.
Aplicação
N-Benzoyl-N-phenylhydroxylamine was used as a complexing agent for studying the dispersive liquid-liquid microextraction based on solidification of floating organic drop (DLLME-SFO) behavior of vanadium (V). It was used as an extractant in the determination of beryllium in natural waters.
Código de classe de armazenamento
11 - Combustible Solids
Classe de risco de água (WGK)
WGK 3
Ponto de fulgor (°F)
Not applicable
Ponto de fulgor (°C)
Not applicable
Equipamento de proteção individual
Eyeshields, Gloves, type N95 (US)
Escolha uma das versões mais recentes:
Já possui este produto?
Encontre a documentação dos produtos que você adquiriu recentemente na biblioteca de documentos.
Xiaoshan Huang et al.
International journal of analytical chemistry, 2018, 8045324-8045324 (2018-08-30)
A new sensitive method for antimony (III) determination by graphite furnace atomic absorption spectrometry (GFAAS) has been developed by using N-benzoyl-N-phenylhydroxylamine (BPHA) and 1-butyl-3-methylimidazolium hexafluorophosphate ([C4mim][PF6]) single drop microextraction. The single drop microextraction (SDMM) system is more competitive compared with
Reaction mechanism of N-benzoyl-N-phenylhydroxylamine with vanadium(IV) in the weakly acidic medium.
Z Nan
Talanta, 52(5), 785-789 (2008-10-31)
The N-benzoyl-N-phenylhydroxylamine(BPHA)-V(IV) system, if kept de-aerated, gives no color reaction. In an open vessel a color reaction does take place. This reaction was studied in solution by spectrophotometry, and the product prepared as solid by scanning thermal analysis, X-ray photoelectron
Tahereh Asadollahi et al.
Talanta, 82(1), 208-212 (2010-08-06)
A novel dispersive liquid-liquid microextraction based on solidification of floating organic drop (DLLME-SFO) for separation/preconcentration of ultra trace amount of vanadium and its determination with the electrothermal atomic absorption spectrometry (ETAAS) was developed. The DLLME-SFO behavior of vanadium (V) using
Jie Li et al.
Talanta, 81(3), 954-958 (2010-03-20)
A simple and rapid analytical method for determining the concentration of rhenium in molybdenite for Re-Os dating was developed. The method used isotope dilution-inductively coupled plasma-mass spectrometry (ID-ICP-MS) after the removal of major matrix elements (e.g., Mo, Fe, and W)
Zhefeng Fan
Analytica chimica acta, 585(2), 300-304 (2007-03-28)
A simple and sensitive method for using electrothermal atomic absorption spectrometry (ET AAS) with Rh as permanent modifier determination of Sb(III) and total Sb after separation and preconcentration by N-benzoyl-N-phenylhydroxylamine (BPHA)-chloroform single drop has been developed. Parameters, such as pyrolysis
Nossa equipe de cientistas tem experiência em todas as áreas de pesquisa, incluindo Life Sciences, ciência de materiais, síntese química, cromatografia, química analítica e muitas outras.
Entre em contato com a assistência técnica