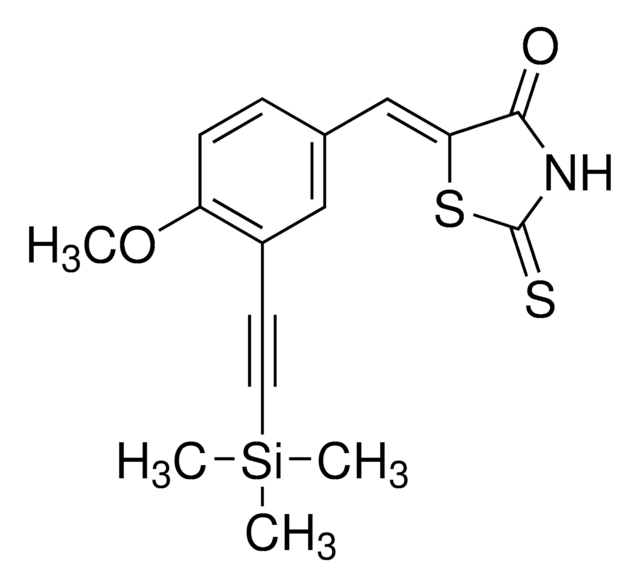
103276
Harmane
98%
Sinônimo(s):
1-Methyl-9H-pyrido[3,4-b]indole, 2-Methyl-β-carboline, Aribine
About This Item
Produtos recomendados
Nível de qualidade
Ensaio
98%
drug control
stupéfiant (France)
pf
235-238 °C (lit.)
solubilidade
methanol: soluble 50mg/ml
cadeia de caracteres SMILES
Cc1nccc2c3ccccc3[nH]c12
InChI
1S/C12H10N2/c1-8-12-10(6-7-13-8)9-4-2-3-5-11(9)14-12/h2-7,14H,1H3
chave InChI
PSFDQSOCUJVVGF-UHFFFAOYSA-N
Informações sobre genes
human ... CYP2D6(1565)
rat ... Gabra2(29706)
Procurando produtos similares? Visita Guia de comparação de produtos
Categorias relacionadas
Descrição geral
- Harmane is a potent tremor-producing β-carboline alkaloid and neurotoxin.
- It is major representative of heterocyclic aromatic amines, a group of mutagenic and carcinogenic substances which are formed in meat from the precursors creatine, creatinine, amino acids and sugars during the heating at high temperatures.
- Blood harmane concentration is elevated in essential tremor, late-life neurological disease.
Aplicação
- Harmane was used in trace level determination of harmane by planar chromatography coupled with (tandem) mass spectrometry.
- It was used to study interactions of norharman and harman with DNA.
- It may be used as matrix for analysis of cyclodextrins and for sulfated oligosaccharides in combination with DHB as co-matrix.
Ações bioquímicas/fisiológicas
Nota de preparo
Palavra indicadora
Warning
Frases de perigo
Declarações de precaução
Classificações de perigo
Acute Tox. 4 Oral - Skin Irrit. 2
Código de classe de armazenamento
11 - Combustible Solids
Classe de risco de água (WGK)
WGK 3
Ponto de fulgor (°F)
Not applicable
Ponto de fulgor (°C)
Not applicable
Equipamento de proteção individual
Eyeshields, Gloves, type N95 (US)
Escolha uma das versões mais recentes:
Já possui este produto?
Encontre a documentação dos produtos que você adquiriu recentemente na biblioteca de documentos.
Os clientes também visualizaram
Artigos
Glycosylation is known to have profound influence on various physiochemical, cellular and biological functions of proteins. Alterations in this modification are known to affect the immune system and have been associated with various pathological states such as cancer, rheumatoid arthritis, and inflammatory diseases.
Glycosylation is known to have profound influence on various physiochemical, cellular and biological functions of proteins. Alterations in this modification are known to affect the immune system and have been associated with various pathological states such as cancer, rheumatoid arthritis, and inflammatory diseases.
Mass Spectrometry of Glycans, method comparison and products
Protocolos
Mass Spectrometry of Glycans
Nossa equipe de cientistas tem experiência em todas as áreas de pesquisa, incluindo Life Sciences, ciência de materiais, síntese química, cromatografia, química analítica e muitas outras.
Entre em contato com a assistência técnica
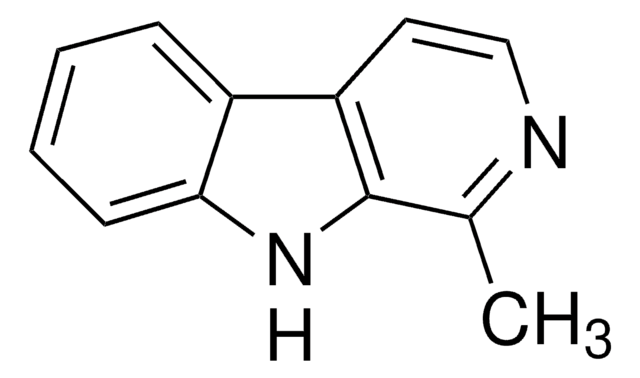
![1,2,3,4-Tetrahydro-9H-pyrido[3,4-b]indole 98%](/deepweb/assets/sigmaaldrich/product/structures/181/460/3d58bc34-1b5c-4295-bbac-3b52085670e8/640/3d58bc34-1b5c-4295-bbac-3b52085670e8.png)
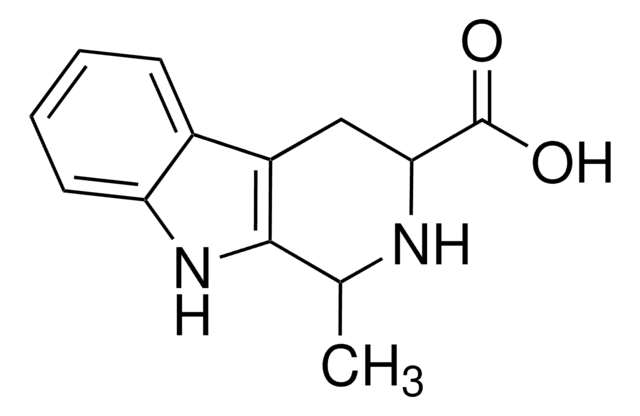
![9H-pyrido[2,3-b]indole AldrichCPR](/deepweb/assets/sigmaaldrich/product/structures/418/992/8c7bac06-11e8-45d7-b863-5d35b582e871/640/8c7bac06-11e8-45d7-b863-5d35b582e871.png)

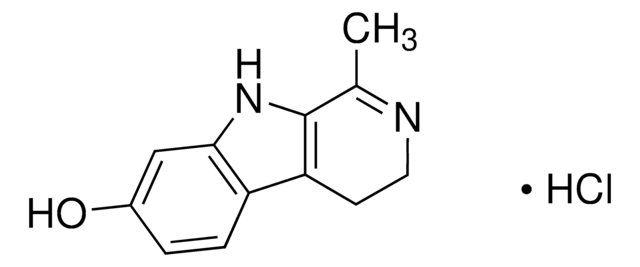
![2,3,4,5-Tetrahydro-1H-pyrido[4,3-b]indole AldrichCPR](/deepweb/assets/sigmaaldrich/product/structures/376/664/07577eb6-6e8c-4237-b8c5-03da4c8e7d88/640/07577eb6-6e8c-4237-b8c5-03da4c8e7d88.png)