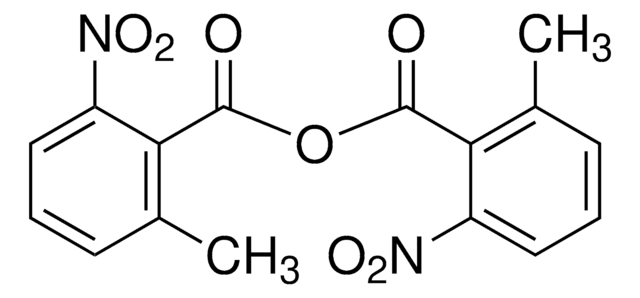
107700
4-(Dimethylamino)pyridine
ReagentPlus®, ≥99%
Synonym(s):
N,N-Dimethylpyridin-4-amine, DMAP
About This Item
Recommended Products
Quality Level
product line
ReagentPlus®
assay
≥99%
form
chips
crystalline powder
flakes
mp
108-110 °C (lit.)
solubility
H2O: 50 mg/mL
storage temp.
room temp
SMILES string
CN(C)c1ccncc1
InChI
1S/C7H10N2/c1-9(2)7-3-5-8-6-4-7/h3-6H,1-2H3
InChI key
VHYFNPMBLIVWCW-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
Application
- To synthesize 3,5-disubstituted 2,6-dicyanoaniline by reacting malononitrile, aldehydes, and β-nitroolefins.
- For the acylation of alcohols with acid anhydrides under auxiliary base- and solvent-free conditions to synthesize corresponding esters.
- In Baylis-Hillman reaction to form carbon-carbon bond by the coupling of an activated alkene with an aldehyde or ketone.
Legal Information
signalword
Danger
Hazard Classifications
Acute Tox. 2 Dermal - Acute Tox. 3 Inhalation - Acute Tox. 3 Oral - Aquatic Chronic 2 - Eye Dam. 1 - Skin Irrit. 2 - STOT SE 1
target_organs
Nervous system
Storage Class
6.1A - Combustible acute toxic Cat. 1 and 2 / very toxic hazardous materials
wgk_germany
WGK 3
flash_point_f
255.2 °F
flash_point_c
124 °C
ppe
Eyeshields, Faceshields, Gloves, type P3 (EN 143) respirator cartridges
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service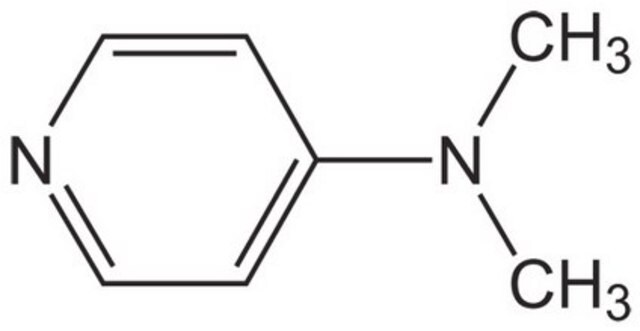







![1,8-Diazabicyclo[5.4.0]undec-7-ene 98%](/deepweb/assets/sigmaaldrich/product/structures/120/564/5b373e23-1624-489c-8efb-692de0f96ffb/640/5b373e23-1624-489c-8efb-692de0f96ffb.png)