Epigenetics
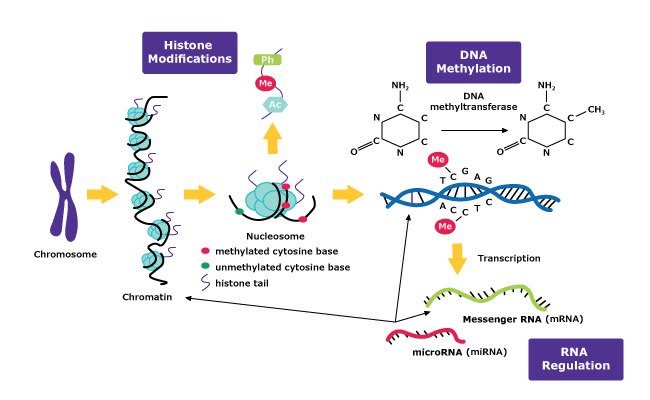
Epigenetic Mechanisms
The field of epigenetics has become an essential area of focus for scientists working on cancer, neurodegenerative disease, and addiction research. Epigenetic mechanisms involve temporarily activating or repressing gene expression. Interestingly, these changes can be passed from generation to generation, even though they do not permanently change the DNA sequence. The three main mechanisms of epigenetics are DNA methylation, histone modification, and RNA regulation.
DNA Methylation
DNA methylation is the most well-known mechanism of epigenetics. It typically involves a methyltransferase enzyme that assists with the addition of a methyl group on the fifth position of cytosine (C5). This addition occurs mainly on cytosine-phosphate- guanine (CpG) dinucleotides. However, non-CpG methylation occurs as well. Analysis of DNA methylation is often performed to help understand gene expression. Examples of this type of analysis include methylation quantification by digestion of DNA with subsequent analysis through HPLC, mass spectrometry, or using sodium bisulfate conversion followed by PCR sequencing and analysis.
Histone Modification
Histone modification is another classic epigenetic mechanism. It involves various ways of altering histones by acetylation, methylation, phosphorylation, and other mechanisms that affect gene expression. Histones are proteins, that along with DNA, make up nucleosomes. Bundles of nucleosomes create the chromatin that make up chromosomes. In general, histone modifications take place at the histone N-terminal tails with high proportions of the amino acids lysine or arginine. One way to study this epigenetic regulation is through the use of chromatin immunoprecipitation (ChIP) assays.
RNA Regulation
Less is known about RNA regulation than the other epigenetic mechanisms. RNA signaling is thought to play a role in epigenetics through regulating chromatin structure. Researchers are investigating how mRNA and specifically non-coding RNA, such as long non-coding RNA, and micro RNA regulate gene expression. Additionally, Chromatin isolation by RNA purification (ChIRP) or RNA immunoprecipitation (RIP) assays can be used to understand the relationship between chromatin and RNA and the role RNA has in epigenetics.
Related Technical Articles
- Epigenetic regulation starts with DNA wound around a set of completely acetylated histones associated with an activated, fully transcribed gene.
- p53 regulates gene expression, cell cycle control and functions as a tumor suppressor. Inactivation of p53 is closely tied to cancer development.
- The Imprint DNA Modification Kit provides the reagents needed for bisulfite conversion and post-modification clean-up of DNA samples in less than 2 hours.
- Epigenetics is a term coined to describe changes that are not mutation based but can still be passed on from generation to generation. Genes that are activated or repressed without any change in DNA sequence are epigenetically controlled. Epigenetic modifications are stable, but potentially reversible alterations in gene expression that occur without permanent changes in DNA sequence.
- Cancer research has revealed that the classical model of carcinogenesis, a three step process consisting of initiation, promotion, and progression, is not complete.
- See All (11)
Related Protocols
- To prevent nonspecific binding of the analyzed protein or extract to the DNA fragment or oligonucleotide, nonspecific competitor nucleic acid is added to the binding reaction.
- The Imprint® Methylated DNA Quantification Kit provides a high-throughput, non-radioactive means of measuring global DNA methylation shifts.
- Chromatin Immunoprecipitation qPCR for studying gene regulation across conditions.
- Protocol for Chromatin Isolation by RNA Purification (ChIRP) Protocol
- The Sigma Imprint Chromatin Immunoprecipitation Kit uses a plate based system to allow rapid ChIP assays in a high throughput format
- See All (5)
Find More Articles and Protocols
To continue reading please sign in or create an account.
Don't Have An Account?