Transform your microscope
CellASIC® ONIX2 Live Cell Imaging System
The ability to grow, observe and manipulate complex cell cultures requires precision control over the cell culture environment. The CellASIC® ONIX2 Microfluidic System transforms your existing microscope into an automated system for advanced live-cell imaging and analysis.
- Dynamic live cell imaging
- Compatible with any inverted microscope
- Small footprint
- User-friendly operation
- Optically clear microfluidic plates for many applications
- Integrated microincubator controller maintains culture environment
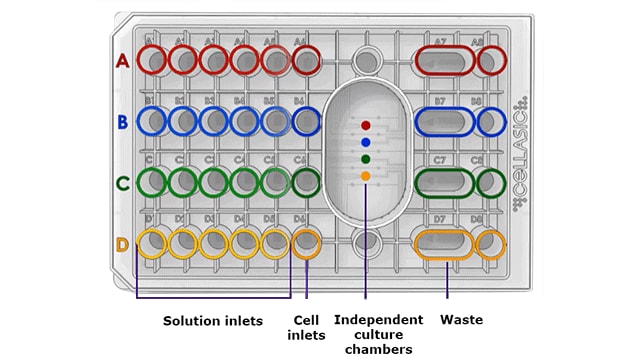
How it works
The CellASIC® ONIX2 system creates a vacuum seal with specialized microfluidic plates and controls perfusion, temperature, and gas environment. Adherent or suspension cells are added directly to the four cell inlet wells of the microfluidic plate. Capillary-driven cell loading automatically loads cells into the culture chambers in a gentle, well-distributed manner. Media or reagents are loaded into the solution inlet wells. Individual microchannels connect each solution to the cell culture chambers in the imaging area of the microfluidic plate. The manifold connecting the plates to the system can be adapted to any inverted microscope.
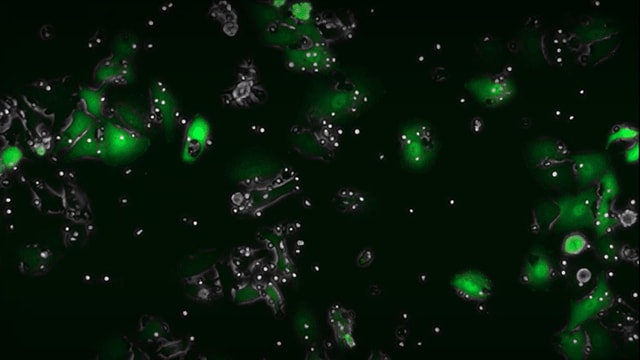
Live cell imaging of NK killing mediated by anti-EGFR using the CellASIC® ONIX2 microfluidic platform.
Applications
Diverse application-specific plate designs give you the flexibility to conduct the culture and assays that yield the most relevant data. Plates are optimized for cells of varying diameters, including mammalian, yeast, and bacterial cells.
- Microscopic analysis of 3D cellular structures
- Chemotaxis/migration in response to chemical gradients
- Real-time and dynamic cellular response to compound addition/withdrawal and changing environmental conditions
- Primary neuron culture over 21 days
- Host-pathogen interactions
- Bacterial single-cell response and biofilm formation

CellASIC® ONIX microfluidic plates
The CellASIC® ONIX microfluidic cell culture plates are designed for optimal cell health, while allowing dynamic live-cell studies with mammalian cells, bacterial cells, and yeast cells.
- Microfluidic volumes allow fast perturbations and exchanges of environmental fluids
- Perfusion microbarriers allow continuous reagent transport without cell stress
- Optical glass bottom and chamber enables high-resolution live-cell imaging
- Flexible design allows multiple independent experiments simultaneously
- Cells have continuous access to nutrients and removal of waste is uninterrupted
Live cell imaging of apoptosis in immune cells
Live cell imaging of hypoxia in cancer cells
Jurkat cells were first loaded onto the CellASIC® ONIX2 System M04T Pad Trap Plate (M04T-01-5PK) and were then incubated with BioTracker NucView® 488 Green Caspase-3 Dye (SCT101) and Annexin V Texas Red for the first 2 hours in complete RPMI-1640 media (SLM-240-B), followed by treatment of 2.5 µg/mL CD-95 antibody (05-201) (experiment group) or no treatment (control) for 8 hours. Time-lapse imaging was taken on cells at a 10-minute interval under 400x magnification using a FITC and Texas-Red filter for the visualization of Caspase 3 activation and Annexin binding, respectively.
A431 epidermal carcinoma cells were incubated 16 hours overnight with 3 µM BioTracker 520 Green Hypoxia Dye (SCT033) in complete DMEM by gravity perfusion at 37°C under normoxic conditions (20% O2) using the CellASIC® ONIX2 System with M04 Switching Plate (M04S-03-5PK). After overnight incubation, cells were perfused with complete DMEM media without dye at 1 psi under hypoxic conditions (0% O2) for 16 hours at 37°C. Fluorescent images were captured over 16 hours. The BioTracker 520 Green Hypoxia Dye increases in fluorescence intensity with decreasing oxygen levels (increased hypoxic conditions).
Live cell imaging of calcium response
Live cell imaging of cardiomyocytes beating
HeLa cells were loaded onto CellASIC® ONIX2 System M04 Switching Plate (M04S-03-5PK). After overnight culturing, Fluo-4AM fluorescent calcium indicator dye was loaded to the HeLa cells and P2Y receptors were activated with ATP to induce increased intracellular calcium. Time-lapse imaging was taken at 1 second intervals.
HL-1 cardiomyoctes were loaded onto the CellASIC® ONIX2 System M04 Switching Plate (M04S-03-5PK). After overnight culturing, Fluo-4AM fluorescent calcium indicator dye was loaded to the cells to visualize spontaneous beating. Real time single cell tracking was also performed on selected cells to visualize calcium flux at the single cell level. Time-lapse imaging was taken at 1 second intervals.
Live cell imaging of immune synapse formation
Live cell imaging of EGFR internalization
T and B cells (Jurkat and Raji, respectively) were first loaded onto the CellASIC® ONIX2 System M04T Pad Trap Plate (M04T-01-5PK). B cells were loaded with SEE (Staphylococcus Enterotoxin E) by perfusion and time lapse imaging was taken at 30 seconds intervals. Square feature in the middle represent the cell trap with 12 micron vertical height restriction. Immune synapse formations can be clearly visualized at single cell level during the entirety of imaging under the cell trap, as the height restriction of the trap prevents the non-adherent suspension lymphocytes from moving out of focal plane.
A549 lung carcinoma cells expressing GFP-EGFR (CLL1141) were loaded onto the CellASIC® ONIX2 System M04 Switching Plate (M04S-03-5PK). After overnight culturing, cells were perfused with anti-EGFR antibody to induce activation of EFGR and its subsequent internalization. Time-lapse imaging was taken at 30 second intervals.
Live cell imaging of mitochondrial depolarization
Live cell imaging of chemotaxis
HeLa cells were first loaded onto the CellASIC® ONIX2 System M04 Switching Plate (M04S-03-5PK). After overnight culturing, cells were loaded with BioTracker 488 Mitochondria dye (SCT136) to visualize the mitochondrial network. Depolarization of mitochondrial network was induced by perfusion of CCCP (carbonyl cyanide 3-chlorophenylhydrazone). Time-lapse imaging was taken at 30 second intervals.
HT-1080 human fibrosarcoma cells were first loaded onto the CellASIC® ONIX2 System M04 Gradient Plate (M04G-02-5PK). After overnight culturing, chemotactic gradient was created by perfusing adjacent parallel flow channels with and without chemoattractant (fetal bovine serum). Time-lapse imaging was taken at 20 minute intervals.
Live cell imaging of autophagosomes
Live cell imaging of tumor spheroids
HT-1080 cells transduced with GFP-LC3 lentiviral particles (17-10193) were first loaded onto the CellASIC® ONIX2 System M04 Switching Plate (M04S-03-5PK). After overnight culturing, cells were perfused with lysosome inhibitor under hypoxic condition to induce autophagy and punctate patterned autophagosomes.
MCF-10A cells were mixed with cold ECM gel and loaded onto pre-chilled CellASIC® ONIX2 System M04 Switching Plate (M04S-03-5PK). After overnight culture, 3D spheroids were imaged at 10 minute intervals for 18 hours.
Leading Biologists Take Control with CellASIC® ONIX Technology
Hear how a lab at Harvard University uses the CellASIC® ONIX system to study how bacteria grow and to identify new targets for antibiotic drugs.
Exploiting the CellASIC® ONIX2 Platform for the Study of Bacterial Cell Biophysics
Learn how a lab at NYU is using the CellASIC® ONIX2 system to study bacterial cell biophysics.
Life is dynamic
Explore our tech articles and protocols for live cell imaging using the CellASIC® ONIX2 system.
To continue reading please sign in or create an account.
Don't Have An Account?