Enzymatic Assay of Malic Dehydrogenase (EC 1.1.1.37)
Description
This procedure may be used for all Malic Dehydrogenase products.
The continuous spectrophotometric rate determination (A340, Light path = 1 cm) is based on the following reaction:
![]()
β-NADH – β-Nicotinamide Adenine Dinucleotide, Reduced form
β-NAD+ – β-Nicotinamide Adenine Dinucleotide, Oxidized Form
MDH – Malic Dehydrogenase
Unit Definition – One unit of malic dehydrogenase will convert 1.0 µmole of Oxaloacetate and β‑NADH to L-Malate and β-NAD+ per minute at pH 7.5 at 25 °C.
Reagents and Equipment Required
Potassium Phosphate, monobasic (P5379)
β-Nicotinamide Adenine Dinucleotide, reduced, disodium salt, hydrate (N8129)
Oxaloacetic Acid (O4126)
Cuvettes and thermostatted spectrophotometer
Precautions
Please consult the Safety Data Sheet for information regarding hazards and safe handling practices.
Preparation Instructions (Storage/Stability)
Use ultrapure water (≥18 MΩcm resistivity at 25 °C) for the preparation of reagents.
Phosphate Buffer (100 mM Potassium Phosphate Buffer, pH 7.5 at 25 °C) – Prepare a 13.6 mg/ml solution in ultrapure water using Potassium Phosphate, monobasic (P5379). Adjust to pH 7.5 at 25 °C with 1.0 M KOH.
β-NADH Solution (0.14 mM β-Nicotinamide Adenine Dinucleotide Solution) – Prepare a 0.11 mg/ml solution in Phosphate Buffer, using β-Nicotinamide Adenine Dinucleotide, reduced, disodium salt, hydrate (N8129). Prepare Fresh.
Note: Concentration of β-NADH is calculated using the molecular weight of 781. This molecular weight is corrected for approximate water content.
OAA Solution (7.6 mM Oxaloacetic Acid) – Immediately prior to use, prepare a 1.0 mg/ml solution Phosphate Buffer, using Oxaloacetic Acid (O4126). Product is not stable once in solution. Prepare Fresh For Each Kinetic Run.
MDH Solution (Malic Dehydrogenase) – Immediately before use, prepare a 0.2–0.5 unit/ml solution in cold Phosphate Buffer. Prepare Fresh.
Procedure
Final Assay Concentrations – In a 3.00 mL Reaction Mixture, the final concentrations are 100 mM Potassium Phosphate, 0.13 mM β-Nicotinamide Adenine Dinucleotide, 0.25 mM Oxaloacetic Acid, and 0.02–0.05 unit of Malic Dehydrogenase.
1. Pipette the following reagent into suitable cuvettes:
2. Equilibrate to 25 °C using a suitably thermostatted spectrophotometer.
3. Then add:
4. Immediately mix by inversion and record the decrease in A340 at 25 °C for ~5 minutes. Using a 1 minute time period and
a minimum of 4 data points, obtain the A340/minute using the maximum linear rate for both the Blank and Test.
Results
Calculations
1. ΔA340/minute = A340/minute (Test) – A340/minute (Blank)
2.
![]()
Where:
3.0 = Total Volume (in milliliters) of Assay
df = Dilution factor
6.22 = Millimolar extinction coefficient of β-NADH at 340 nm
VE = Volume (in milliliters) of enzyme used
3.
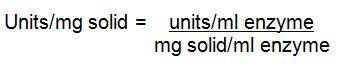
Reference
To continue reading please sign in or create an account.
Don't Have An Account?