V0627
Valinomycin
≥98% (TLC), ≥90% (HPLC)
Synonym(s):
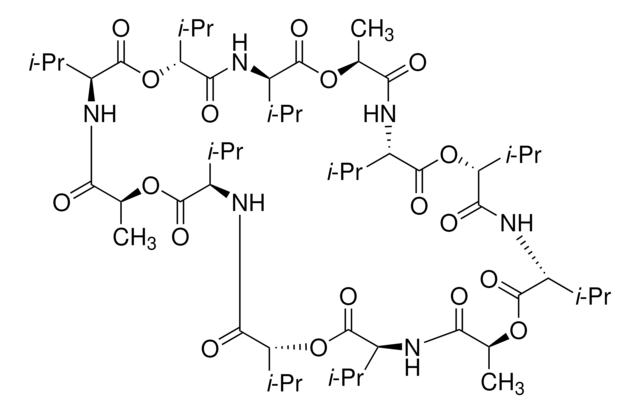
Cyclo(L-Val-D-HyIva-D-Val-L-Lac-)3: HyIva = α-Hydroxyisovaleric acid, Lac = Lactic acid
About This Item
Recommended Products
Quality Level
assay
≥90% (HPLC)
≥98% (TLC)
form
powder
storage condition
(Keep container tightly closed in a dry and well-ventilated place.)
color
white
solubility
DMSO: ≥50 mg/mL
H2O: insoluble
antibiotic activity spectrum
Gram-positive bacteria
parasites
viruses
mode of action
cell membrane | interferes
storage temp.
2-8°C
SMILES string
CC(C)[C@@H]1NC(=O)[C@H](C)OC(=O)[C@@H](NC(=O)[C@H](OC(=O)[C@@H](NC(=O)[C@H](C)OC(=O)[C@H](NC(=O)[C@H](OC(=O)[C@@H](NC(=O)[C@H](C)OC(=O)[C@H](NC(=O)[C@H](OC1=O)C(C)C)C(C)C)C(C)C)C(C)C)C(C)C)C(C)C)C(C)C)C(C)C
InChI
1S/C54H90N6O18/c1-22(2)34-49(67)73-31(19)43(61)55-38(26(9)10)53(71)77-41(29(15)16)47(65)59-36(24(5)6)51(69)75-33(21)45(63)57-39(27(11)12)54(72)78-42(30(17)18)48(66)60-35(23(3)4)50(68)74-32(20)44(62)56-37(25(7)8)52(70)76-40(28(13)14)46(64)58-34/h22-42H,1-21H3,(H,55,61)(H,56,62)(H,57,63)(H,58,64)(H,59,65)(H,60,66)/t31-,32-,33-,34-,35+,36+,37-,38-,39-,40+,41+,42+/m0/s1
InChI key
FCFNRCROJUBPLU-RPUZOQEISA-N
Looking for similar products? Visit Product Comparison Guide
General description
Application
- to equilibrate intracellular pH and extracellular pH of 5 (and 6-)-carboxyfluorescein succinimidyl ester (cFSE) in Escherichia coli strain B23
- as an ionophore to equilibrate intracellular pH and extracellular pH of lactic acid bacteria (LAB) strains
- to establish the desired pH for acid-base transition and formation of K+-valinomycin diffusion potential
Biochem/physiol Actions
Packaging
Other Notes
signalword
Danger
hcodes
Hazard Classifications
Acute Tox. 1 Dermal - Acute Tox. 1 Oral
Storage Class
6.1A - Combustible acute toxic Cat. 1 and 2 / very toxic hazardous materials
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Faceshields, Gloves, type P3 (EN 143) respirator cartridges
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service