R7632
Retinol
synthetic, ≥95% (HPLC), (Powder or Powder with Lumps)
Synonym(s):
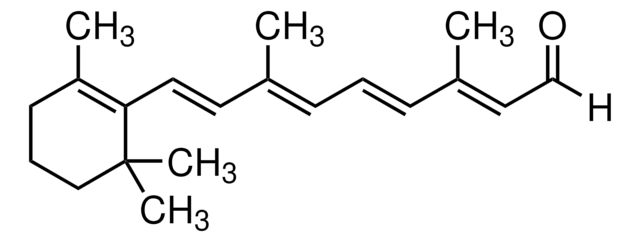
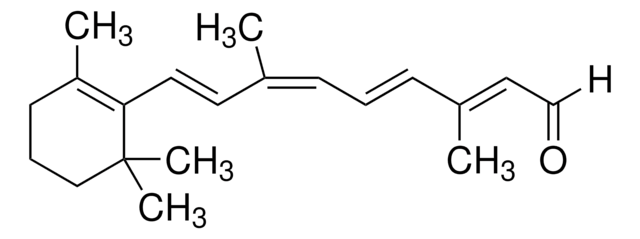
Axerophthol, Vitamin A, Vitamin A alcohol, Vitamin A1, all-trans-3,7-Dimethyl-9-(2,6,6-trimethyl-1-cyclohexen-1-yl)-2,4,6,8-nonatetraen-1-ol
About This Item
Recommended Products
biological source
synthetic
Quality Level
assay
≥95% (HPLC)
form
(Powder or Powder with Lumps)
specific activity
~2700 U/mg
technique(s)
HPLC: suitable
color
yellow to very dark yellow, to Very Dark Orange
mp
61-63 °C (lit.)
shipped in
dry ice
storage temp.
−20°C
SMILES string
CC1(C)C(/C=C/C(C)=C/C=C/C(C)=C/CO)=C(C)CCC1
InChI
1S/C20H30O/c1-16(8-6-9-17(2)13-15-21)11-12-19-18(3)10-7-14-20(19,4)5/h6,8-9,11-13,21H,7,10,14-15H2,1-5H3/b9-6+,12-11+,16-8+,17-13+
InChI key
FPIPGXGPPPQFEQ-OVSJKPMPSA-N
Looking for similar products? Visit Product Comparison Guide
General description
Application
- for the synthesis of all-trans-retinoic acid in HepG2 cells
- to study the effect of retinol on the growth of murine normal colonic cells cultured as organoids
- as a standard for determination of vitamin A in cells
- as a component of chemically defined medium for testis organ culture and spermatogenesis in vitro.
Biochem/physiol Actions
Packaging
signalword
Danger
hcodes
Hazard Classifications
Aquatic Chronic 4 - Eye Irrit. 2 - Repr. 1B - Skin Sens. 1
Storage Class
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
wgk_germany
WGK 1
ppe
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
All-trans retinoic acid (RA, ATRA) is a pleiotropic activation factor that regulates genes associated with normal vertebrate cellular processes such as cell differentiation, cell proliferation, apoptosis, and embryonic development.
All-trans retinoic acid (RA, ATRA) is a pleiotropic activation factor that regulates genes associated with normal vertebrate cellular processes such as cell differentiation, cell proliferation, apoptosis, and embryonic development.
Protocols
Separation of δ-Tocopherol, analytical standard; Retinyl acetate; (+)-γ-Tocopherol; α-Tocopherol, ≥95.5%; Retinol; Vitamin A (acetate), meets USP testing specifications.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service