General description
Horseradish peroxidase is isolated from horseradish roots (Amoracia rusticana) and belongs to the ferroprotoporphyrin group of peroxidases. HRP is a single chain polypeptide containing four disulfide bridges. It is a glycoprotein containing 18% carbohydrate. The carbohydrate composition consists of galactose, arabinose, xylose, fucose, mannose, mannosamine, and galactosamine depending upon the specific isozyme. Its molecular weight (~44 kDa) includes the polypeptide chain (33,890 Daltons), hemin plus Ca2+ (~700 Daltons), and carbohydrate (~9,400 Daltons). At least seven isozymes of HRP exist. The isoelectric point for horseradish peroxidase isozymes ranges from 3.0 - 9.0.
Application
Horseradish peroxidase (HRP) is isolated from horseradish roots (Amoracia rusticana). It is used in biochemistry applications such as western blots, ELISA and Immunohistochemistry. Horseradish peroxidase is used to amplify a weak signal and increase detectability of a target molecule, such as a protein. Product P6140 has been used to detect low density lipoprotein (LDL).
The enzyme from Sigma has been used while assessing the skin sensitization potential of pro-haptens. It has also been used to show that peroxidase (PO) activity and its heat stability correlate with the availability of free Ca2+ ions.
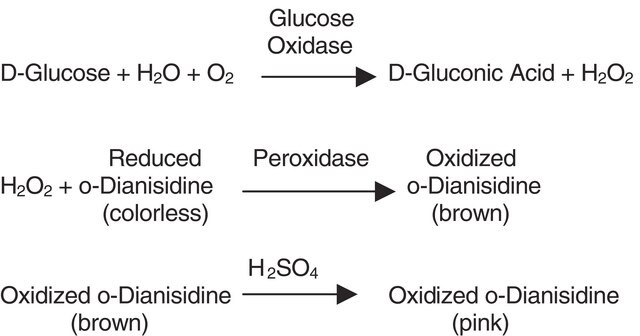
Biochem/physiol Actions
HRP readily combines with hydrogen peroxide (H2O2) and the resultant [HRP-H2O2] complex can oxidize a wide variety of hydrogen donors. The optimal pH is 6.0-6.5 and the enzyme is most stable in the pH range of 5.0-9.0. HRP can be conjugated to antibodies by several different methods that include the use of glutaraldehyde, periodate oxidation, disulfide bonds, and also via amino- and thiol-directed cross-linkers. It is smaller and more stable than the enzyme labels, β-galactosidase and alkaline phosphatase. Hence, it is the most desired label. Also, its glycosylation leads to lower non-specific binding. It is also used for the determination of glucose and peroxides in solution. Sodium azide, cyanide, L-cystine, dichromate, ethylenethiourea, hydroxylamine, sulfide, vanadate, p-aminobenzoic acid, and Cd2+, Co2+, Cu2+, Fe3+, Mn2+, Ni2+, Pb2+ ions are found to inhibit its enzyme activity.
When incubated with a substrate, horseradish peroxidase produces a coloured, fluorimetric, or luminescent derivative of the labeled molecule, allowing quantification. Horseradish peroxidase has been shown to slightly reduce the level of inhibition in a cydAB mutant.
Unit Definition
One pyrogallol unit will form 1.0 mg purpurogallin from pyrogallol in 20 sec at pH 6.0 at 20 °C.
Physical form
Crystalline suspension in 3.2 M (NH4)2SO4 solution containing potassium phosphate buffer, pH 6.0
Preparation Note
Water may be used to dilute suspension if needed.
Analysis Note
Preliminary studies indicate the presence of two basic and no acidic isoenzymes
The RZ (Reinheitszahl) is the absorbance ratio A403/A275 determined at 0.5-1.0 mg/ml in deionized water. It is a measure of hemin content, not enzymatic activity. Even preparations with high RZ may have low enzymatic activity.