K3125
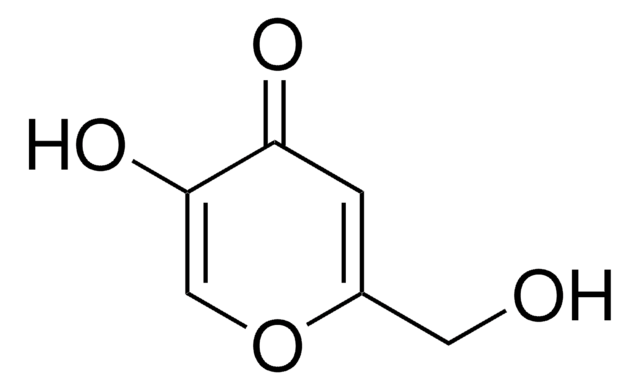
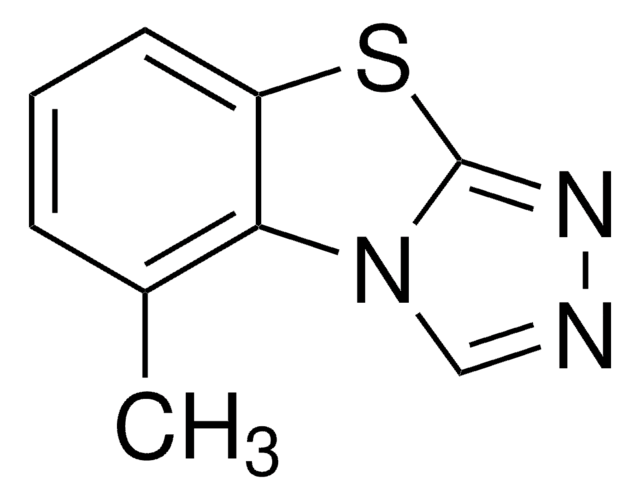
Kojic acid
≥98.5% (HPLC), powder, tyrosinase inhibitor
Synonym(s):
2-Hydroxymethyl-5-hydroxy-γ-pyrone, 5-Hydroxy-2-hydroxymethyl-4H-4-pyranone
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C6H6O4
CAS Number:
Molecular Weight:
142.11
Beilstein/REAXYS Number:
120895
EC Number:
MDL number:
UNSPSC Code:
12352106
PubChem Substance ID:
NACRES:
NA.77
Recommended Products
Product Name
Kojic acid,
assay
≥98.5% (HPLC)
Quality Level
form
powder
mp
152-155 °C (lit.)
SMILES string
OCC1=CC(=O)C(O)=CO1
InChI
1S/C6H6O4/c7-2-4-1-5(8)6(9)3-10-4/h1,3,7,9H,2H2
InChI key
BEJNERDRQOWKJM-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Application
Kojic acid has been used:
- as an inhibitor of tyrosinase in guinea pigs pigmented hyperopic (PH)
- as a reference inhibitor standard for screening tyrosinase inhibition
- as a positive control for inhibition of tyrosinase in B16F10 melanoma cells
Biochem/physiol Actions
Kojic acid is derived from some fungal species such as, Aspergillus, Acetobacter and Penicillium.. It halts melanin synthesis by inhibiting tyrosinase enzyme. It is used in the preparation of skin whitening cosmetics. However, kojic acid usage is minimal in cosmetics, as it induces skin irritation by its unstability and cytotoxic nature during long storage. It is an antioxidant and elicits radioprotective effects on chelating with manganese and zinc.
Tyrosinase inhibitor.
Storage Class
11 - Combustible Solids
wgk_germany
WGK 1
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, type N95 (US)
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Effects of the Tyrosinase-Dependent Dopaminergic System on Refractive Error Development in Guinea Pigs
Jiang L, et al.
Investigative Ophthalmology & Visual Science, 59(11), 4631-4638 (2018)
Ka-Heng Lee et al.
European journal of medicinal chemistry, 44(8), 3195-3200 (2009-04-11)
A series of 46 curcumin related diarylpentanoid analogues were synthesized and evaluated for their anti-inflammatory, antioxidant and anti-tyrosinase activities. Among these compounds 2, 13 and 33 exhibited potent NO inhibitory effect on IFN-gamma/LPS-activated RAW 264.7 cells as compared to L-NAME
Kojic acid and its manganese and zinc complexes as potential radioprotective agents
Emami S, et al.
Bioorganic & Medicinal Chemistry Letters, 17(1), 45-48 (2007)
Xiao Hu et al.
Journal of natural products, 75(1), 82-87 (2011-12-15)
Two novel 2-arylbenzofuran dimers, morusyunnansins A and B (1 and 2), two new biflavonoids, morusyunnansins C and D (3 and 4), two new flavans, morusyunnansins E and F (5 and 6), and four known flavans (7-10) were isolated from the
Wei Yi et al.
European journal of medicinal chemistry, 46(9), 4330-4335 (2011-07-23)
Melanin play a major role in human skin protection and their biosynthesis is vital. Due to their color, they contribute to the skin pigmentation. Tyrosinase is a key enzyme involved in the first stage of melanin biosynthesis, it catalyzes the
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service