H8541
Transforming Growth Factor-β1 human
≥95% (SDS-PAGE), recombinant, expressed in HEK 293 cells, lyophilized powder, suitable for cell culture
Synonym(s):
TGF-β1
About This Item
Recommended Products
product name
Transforming Growth Factor-β1 human, TGF-β1, Xeno-free, recombinant, expressed in HEK 293 cells, suitable for cell culture
biological source
human
Quality Level
recombinant
expressed in HEK 293 cells
assay
≥95% (SDS-PAGE)
form
lyophilized powder
potency
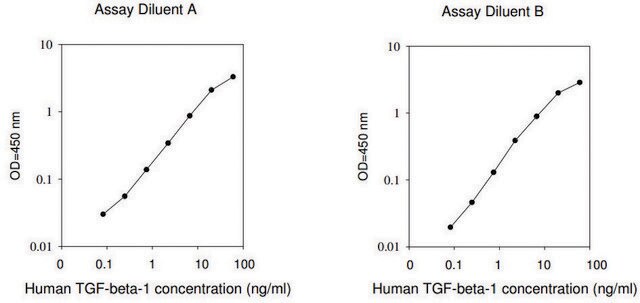
≤0.5 ng/mL EC50
quality
endotoxin tested
mol wt
dimer 25 kDa (non-glycosylated)
packaging
pkg of 5 μg
storage condition
avoid repeated freeze/thaw cycles
technique(s)
cell culture | mammalian: suitable
impurities
≤1 EU/μg
UniProt accession no.
storage temp.
-10 to -25°C
Gene Information
human ... TGFB1(7040)
Looking for similar products? Visit Product Comparison Guide
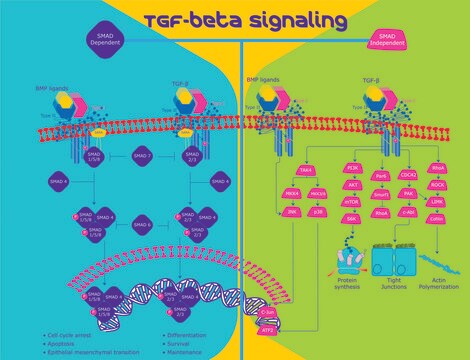
General description
Application
- to study the expression of PDGFRB (Platelet derived growth factor receptor β) in association with transforming growth factor-β signaling.
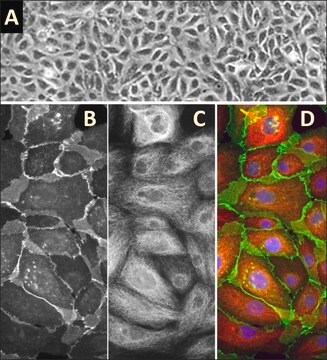
- to stimulate lung fibrosis in human fetal lung fibroblast cell line in order to further measure the in-plane elasticity of live cell layers using a pressure sensor embedded microfluidic device.
- to study the effect of TGF-β on HTRA1 (high-temperature requirement A1)-affected Wnt target genes.
Biochem/physiol Actions
Physical form
Analysis Note
signalword
Danger
hcodes
Hazard Classifications
Eye Dam. 1 - Skin Corr. 1B
Storage Class
8A - Combustible corrosive hazardous materials
wgk_germany
WGK 2
flash_point_f
Not applicable
flash_point_c
Not applicable
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service