H6908
Anti-HA antibody produced in rabbit
affinity isolated antibody, buffered aqueous solution
Synonym(s):
Anti Ha, Anti Ha Antibody, Anti-Ha Antibody, HA Antibody - Anti-HA antibody produced in rabbit, Ha Antibody, Ha Antibody Sigma, Anti-HA
About This Item
Recommended Products
biological source
rabbit
conjugate
unconjugated
antibody form
affinity isolated antibody
antibody product type
primary antibodies
clone
polyclonal
form
buffered aqueous solution
technique(s)
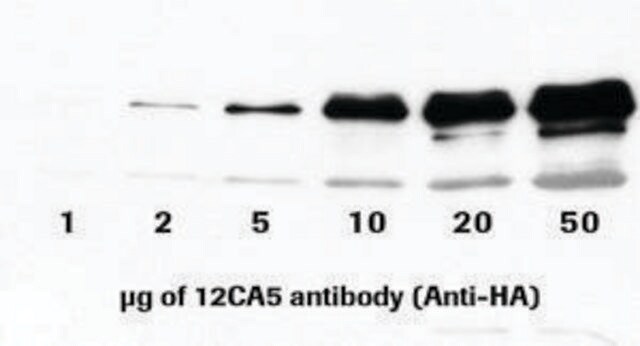
immunoprecipitation (IP): 2.5-4 μg/test using HA-tagged fusion protein from cell lysates
indirect immunofluorescence: 10-20 μg/mL using HA-tagged fusion protein transfected cells
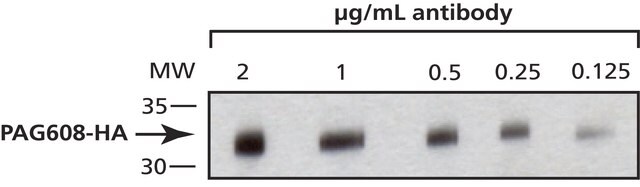
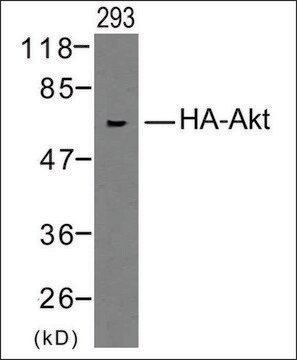
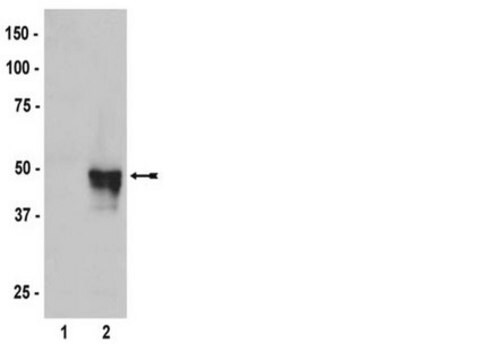
western blot: 0.5-0.8 μg/mL using HA-tagged fusion protein transfected cell extracts
application(s)
research pathology
shipped in
dry ice
storage temp.
−20°C
target post-translational modification
unmodified
General description
Influenza hemagglutinin protein is a nonapeptide derived from the major spike membrane glycoprotein of the human influenza virus. This strain specific glycoprotein is a homotrimer of 84 kDa monomers. Each monomers contain two disulfide-linked subunits: HA1 and HA2.
Anti-HA is produced in rabbit using a synthetic peptide corresponding to amino acid residues of the human Influenza hemagglutinin (HA), conjugated to KLH. The antibody is affinity-purified on the immobilized immunizing peptide.
Anti-HA antibody is specific for N- or C-terminal HA-tagged fusion proteins. The product may detect cross-reacting bands in certain mammalian cells. The immunizing HA peptide (Product No.I2149) specifically inhibits the staining of the HA-tagged protein band.
Specificity
Immunogen
Application
It is also suitable for indirect immunofluorescence (10-20μg/mL using HA-tagged fusion protein transfected cells), immunoprecipitation (2.5-4μg/test using HA-tagged fusion protein from cell lysates), and western blot (0.5-0.8μg/mL using HA-tagged fusion protein transfected cell extracts).
Biochem/physiol Actions
The HA gene is often used as an affinity tag for target proteins in recombinant expression vector systems. The HA-tagged proteins produced from such systems yield stable fusion products that do not interfere with the function and distribution of target proteins. Anti-HA antibody may detect cross-reacting bands in certain mammalian cells. The immunizing HA peptide (Product No.I2149) specifically inhibits the staining of the HA-tagged protein band. The nucleic acid sequence encoding the HA peptide has been incorporated into various expression plasmids adjacent to the cloning site thus enabling the cloning and expression of HA-tagged fusion protein. Such fusion proteins may be expressed in cells of various organisms: bacteria, yeast, insects and mammals. In the fusion protein, the HA sequence may serve as a recognition target for specific antibodies thus enabling detection, subcellular localization, characterization, quantification, functional analysis and affinity purification of the HA-tagged protein and associated bound proteins. Insertion of the HA epitope in different regions of a cellular protein followed by examination of the immunoreactivity of the epitope in intact and in permeabilized cells is useful for studying the cellular expression levels, topology and functional activity of the tagged protein. Antibody mediated detection of the HA tag obviates the need for time consuming generation of antibody specific for newly identified, low abundance, unstable, difficult to purify, cross-reactive or poorly immunogenic proteins.
Physical form
Storage and Stability
For extended storage freeze in working aliquots. Repeated freezing and thawing is not recommended. If slight turbidity occurs upon prolonged storage, clarify the solution by centrifugation before use. Solutions at working dilution should be discarded if not used within 12 hours.
Disclaimer
Not finding the right product?
Try our Product Selector Tool.
Storage Class
12 - Non Combustible Liquids
wgk_germany
WGK 2
flash_point_f
Not applicable
flash_point_c
Not applicable
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Related Content
Protein and nucleic acid interaction reagents and resources for investing protein-RNA, protein-DNA, and protein-protein interactions and associated applications.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service