C4396
Carbonic Anhydrase I from human erythrocytes
Synonym(s):
Carbonate Dehydratase, Carbonate Hydrolyase, Carbonic Anhydrase Isozyme I
Sign Into View Organizational & Contract Pricing
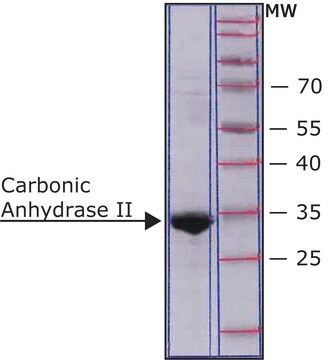
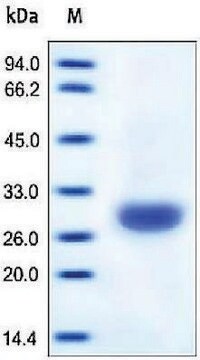
All Photos(1)
About This Item
Recommended Products
form
powder
Quality Level
specific activity
100-500 W-A units/mg protein
mol wt
30 kDa
color
white
pI
~6.6
UniProt accession no.
storage temp.
−20°C
Gene Information
human ... CA1(759)
Looking for similar products? Visit Product Comparison Guide
Application
Carbonic anhydrase from human erythrocytes (HCA) has been used to study the molten-globule state of carbonic anhydrase (CA). Chaperone-like α-crystallin binds to this state of the enzyme and prevents its aggregation. The enzyme from sigma has been used for the analysis of thermodynamic stability of the enzyme. Furthermore, its clinical significance has been evaluated in human non-small cell lung cancer.
Biochem/physiol Actions
Carbonic anhydrase is a zinc metalloenzyme that has a molecular weight of approximately 30,000 Da. The enzyme catalyzes the hydration of carbon dioxide to carbonic acid. It is involved in vital processes such as pH and CO2 homeostasis, transport of bicarbonate and CO2, biosynthetic reactions, bone resorption, calcification, and tumorigenicity. Therefore, this enzyme is an important target for inhibitors with clinical applications in various pathologies such as glaucoma, epilepsy and Parkinson′s disease.
Unit Definition
One Wilbur-Anderson (W-A) unit will cause the pH of a 0.02 M Trizma buffer to drop from 8.3 to 6.3 per min at 0 °C. (One W-A unit is essentially equivalent to one Roughton-Booth unit.)
Preparation Note
chromatographically
purified.
purified.
inhibitor
Product No.
Description
Pricing
signalword
Danger
hcodes
pcodes
Hazard Classifications
Resp. Sens. 1
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
K Rajaraman et al.
The Journal of biological chemistry, 271(44), 27595-27600 (1996-11-01)
alpha-Crystallin, a multimeric protein, exhibits chaperone-like activity in preventing aggregation of several proteins. We have studied the chaperone-like activity of alpha-crystallin toward heat-induced aggregation of bovine and human carbonic anhydrase. Human carbonic anhydrase aggregates at 60 degrees C, while bovine
Whei Ling Chiang et al.
Cancer letters, 188(1-2), 199-205 (2002-10-31)
This study was designed to elucidate the possible relationship between the expression of cytosolic carbonic anhydrase (CA) and non-small cell lung cancer (NSCLC). The activity and protein expression patterns of carbonic anhydrase I (CAI) and II (CAII) of 70 NSCLC
Daumantas Matulis et al.
Biochemistry, 44(13), 5258-5266 (2005-03-30)
ThermoFluor (a miniaturized high-throughput protein stability assay) was used to analyze the linkage between protein thermal stability and ligand binding. Equilibrium binding ligands increase protein thermal stability by an amount proportional to the concentration and affinity of the ligand. Binding
Pavel V Shliaha et al.
Journal of proteome research, 12(6), 2323-2339 (2013-03-22)
qTOF mass spectrometry and traveling wave ion mobility separation (TWIMS) hybrid instruments (q-TWIMS-TOF) have recently become commercially available. Ion mobility separation allows an additional dimension of precursor separation inside the instrument, without incurring an increase in instrument time. We comprehensively
Qiang Jin et al.
Biosensors & bioelectronics, 83, 193-199 (2016-04-30)
A near-infrared fluorescent probe (DDAB) for highly selective and sensitive detection of carboxylesterase 2 (CE2) has been designed, synthesized, and systematically studied both in vitro and in vivo. Upon addition of CE2, the ester bond of DDAB could be rapidly
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service