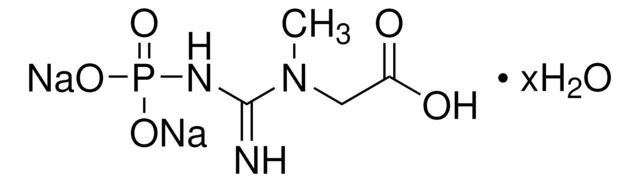
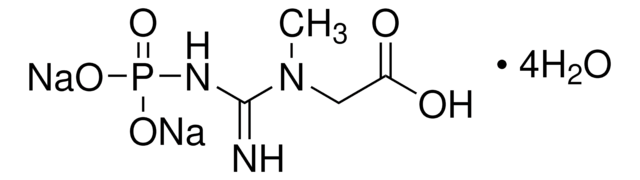
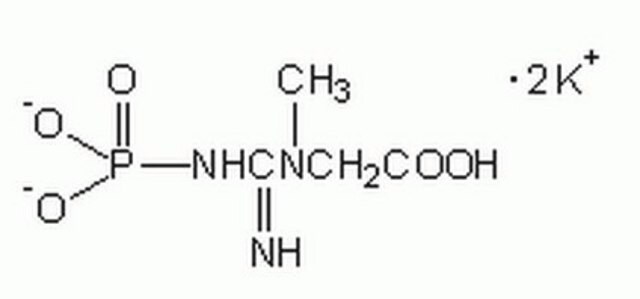
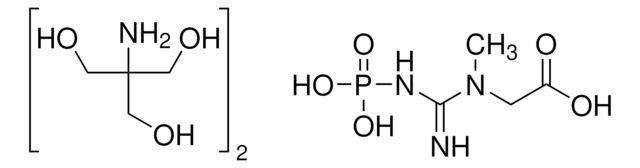
C3755
Creatine Phosphokinase from rabbit muscle
Type I, salt-free, lyophilized powder, ≥150 units/mg protein
Synonym(s):
ATP: Creatine N-Phosphotransferase, CPK, Creatine Kinase, Phosphocreatine phosphokinase
About This Item
Recommended Products
type
Type I
Quality Level
form
salt-free, lyophilized powder
specific activity
≥150 units/mg protein
mol wt
80-86.2 kDa
solubility
0.25 M glycyl-glycine, pH 7.4: soluble 5.0 mg/mL, clear, colorless to slightly yellow
foreign activity
ATPase ≤0.01%
Lactic dehydrogenase, hexokinase, myokinase and pyruvate kinase ≤0.001%
shipped in
wet ice
storage temp.
−20°C
Looking for similar products? Visit Product Comparison Guide
Application
Creatine Phosphokinase is a dimer composed predominantly of the skeletal muscle derived homodimer (MM). CK also exists as a heterodimer (MB) particularly in the myocardium. CK derived from brain tissue consists mainly of the brain source homodimer (BB). The amino acid sequences of the M chain and B chains are about 80% homologous. From the sequence, the molecular weight of the M chain is 43,112.
E1%(280)= 8.76
pH Optimum: pH 8.8-9.0 for the forward reaction and pH 6.0-7.0 for the reverse reaction.
CK is a cellular enzyme with a wide tissue distribution. Its physiological role is associated with ATP generation for contractile or transport systems. Increased levels of CK are associated with myocardial infarction, muscular dystrophy, hyperthyroidism, pulmonary infarction and cerebrovascular disease. Variations in relative isozyme distribution can provide additional information in the diagnosis of these conditions.
Substrates: Creatine, N-ethylglycocyamine and glyocyamine have been shown to act as substrates for CK. CK is very specific for ATP/ADP.
Inhibitors: ADP is a strong inhibitor of the forward reaction competing with ATP. Divalent cations such as Ca2+ (Ki=4.5 mM), Zn2+ and Cu2+ inhibit CK by competing with Mg2+. Other inhibitors include acetate, acetylsalicylic acid, adenosine, p-aminosalicylic acid, AMP, benzoic acid, bicarbonate, bromide, chloride, p-Chloromercuribenzoic acid, ethylene oxide, 2,4-fluorodinitrobenzene, iodide, malonic acid, NAD, nitrate, phosphate, pyrophosphate, salicylic acid, sulfate, sulfite, thyroxine, trichloroacetate, L-triiodothyroxine, L-triiodothyronine, and tripolyphosphate.
Caution
Unit Definition
Preparation Note
Analysis Note
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, type N95 (US)
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service