C0692
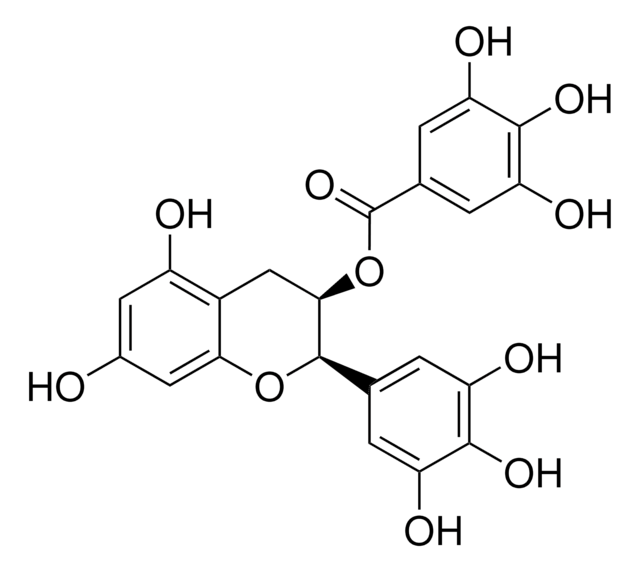
(−)-Catechin gallate
≥98% (HPLC), from green tea
Synonym(s):
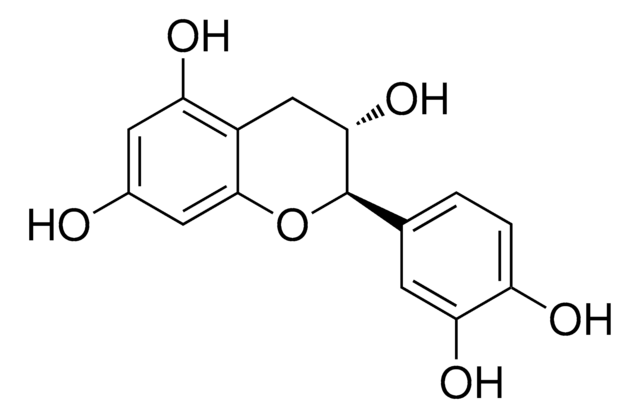
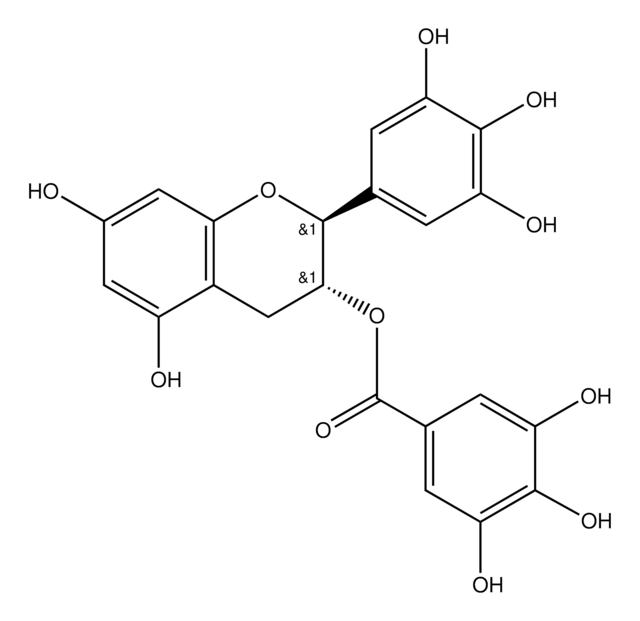
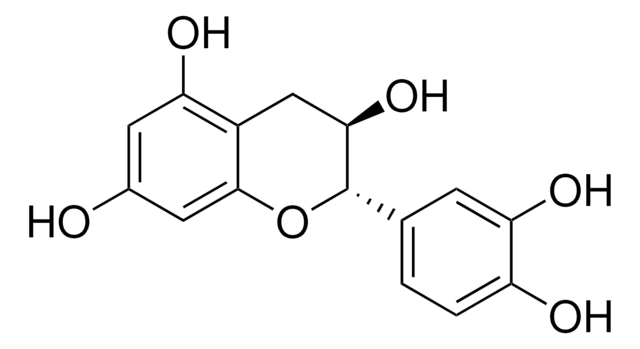
(2S,3R)-2-(3,4-Dihydroxyphenyl)-3,4-dihydro-1(2H)-benzopyran-3,5,7-triol 3-(3,4,5-trihydroxybenzoate)
About This Item
Recommended Products
biological source
green tea
Quality Level
assay
≥98% (HPLC)
form
powder
application(s)
metabolomics
vitamins, nutraceuticals, and natural products
storage temp.
2-8°C
SMILES string
Oc1cc(O)c2C[C@@H](OC(=O)c3cc(O)c(O)c(O)c3)[C@@H](Oc2c1)c4ccc(O)c(O)c4
InChI
1S/C22H18O10/c23-11-6-14(25)12-8-19(32-22(30)10-4-16(27)20(29)17(28)5-10)21(31-18(12)7-11)9-1-2-13(24)15(26)3-9/h1-7,19,21,23-29H,8H2/t19-,21+/m1/s1
InChI key
LSHVYAFMTMFKBA-CTNGQTDRSA-N
Gene Information
human ... BACE1(23621)
Looking for similar products? Visit Product Comparison Guide
General description
Application
- to examine the potency of catechins containing galloyl moiety in inhibiting the activity of human immunodeficiency virus-1 (HIV-1)integrase
- to analyze the anti-oxidant and anti-viral properties of Pseudopiptadenia contorta and the commercial quebracho extracts in a herpes simplex virus type 1 strain, resistant to acyclovir
- to study the effects of polyphenols on the biochemistry of human spermatozoa and the associated limitations of their use in gamete preservation
Biochem/physiol Actions
signalword
Warning
hcodes
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
target_organs
Respiratory system
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
Antioxidants protect biological systems from oxidative damage produced by oxygen-containing free radicals and from redoxactive transition metal ions such as iron, copper, and cadmium.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service