C8571
Cathepsin B from human liver
buffered aqueous solution, ≥1,500 units/mg protein (E1%/280)
Synonym(s):
Cathepsin B1
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
CAS Number:
MDL number:
UNSPSC Code:
12352200
NACRES:
NA.32
Recommended Products
biological source
human liver
Quality Level
form
buffered aqueous solution
specific activity
≥1,500 units/mg protein (E1%/280)
UniProt accession no.
shipped in
dry ice
storage temp.
−70°C
Gene Information
human ... CTSB(1508)
General description
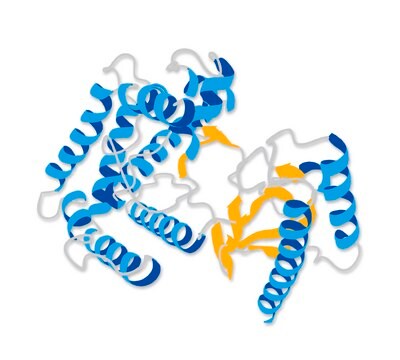
Cathepsin B gene is mapped to human chromosome 8p23.1. It is a lysosomal cysteine with 252 amino acids distributed in two chains and is homologous to rat liver sequence. It has disc shaped structure with two domains namely the left-hand ′L′ domain at the amino terminal and right hand ′R′ domain at the carboxyl-terminal.
Application
Cathepsin B from human liver has been used:
- as a positive control in the screening Cathepsin B inhibitor assay
- in wall extension (expansin activity) assay
- in microinjection human foreskin fibroblasts to test its effect on apoptosis induction
Biochem/physiol Actions
Cathepsin B from human liver is highly expressed in the esophageal adenocarcinoma (EAC). It reduces hepatic inflammation and fibrogenesis and may serve as a potential candidate for treating liver diseases.
Cathepsin B has been found to cleave procaspase 1 and procaspase 11 and to induce apoptosis in digitonin-permeabilized cells. Translocation of cathepsin B from the cytoplasm to the nucleus contributes to bile salt induced apoptosis of rat hepatocytes. Levels of cathepsin B in PC12 cells significantly decrease 12 to 24 hours after apoptosis is induced.
Found to cleave procaspase 1 and procaspase 11 and to induce apoptosis in digitonin-permeabilized cells.
Packaging
Package size based on protein content
Unit Definition
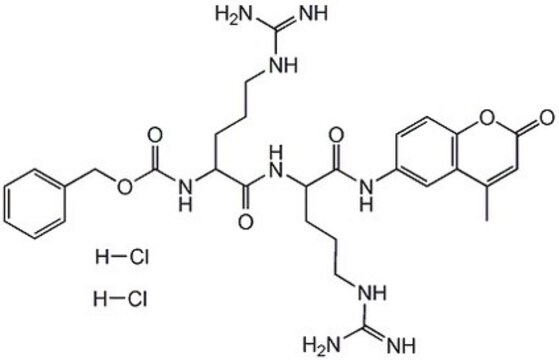
One unit will liberate 1 nanomole of 7-amino-4-methylcoumarin from Z-Arg-Arg 7-amido-4-methylcoumarin per min at pH 6.0 at 40 °C.
Physical form
Solution in 50 mM sodium acetate, pH 5.0, with 1 mM EDTA.
Inhibitor
Product No.
Description
Pricing
Related product
Storage Class
10 - Combustible liquids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Prevention of free fatty acid-induced hepatic lipotoxicity by 18beta-glycyrrhetinic acid through lysosomal and mitochondrial pathways
Wu X, et al.
Hepatology, 47(6), 1905-1915 (2008)
Amino acid sequence of human liver cathepsin B
Ritonja A, et al.
Febs Letters, 181(1), 169-172 (1985)
Genome-wide catalogue of chromosomal aberrations in barrett's esophagus and esophageal adenocarcinoma: a high-density single nucleotide polymorphism array analysis
Gu J, et al.
Cancer Prevention Research (Philadelphia, Pa.), 3(9), 1176-1186 (2010)
The refined 2.15 AX-ray crystal structure of human liver cathepsin B: the structural basis for its specificity.
Musil DJ, et al.
The Embo Journal, 10(9), 2321-2330 (1991)
Cathepsin B inactivation attenuates hepatic injury and fibrosis during cholestasis
Canbay A, et al.
The Journal of Clinical Investigation, 112(2), 152-159 (2003)
Articles
This procedure applies to all products that have a specification for Cathepsin B activity determined by the liberation of 7-amino-4-methylcoumarin from Z-Arg-Arg 7-amido-4-methylcoumarin.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service