G4251
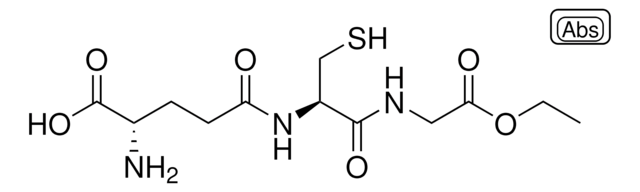
L-Glutathione reduced
≥98.0%
Synonym(s):
Glutathione-SH, γ-L-Glutamyl-L-cysteinyl-glycine, GSH
About This Item
Recommended Products
Product Name
L-Glutathione reduced, ≥98.0%
Quality Level
assay
≥98.0%
form
powder
color
white
mp
192-195 °C (dec.) (lit.)
solubility
water: 50 mg/mL, clear, colorless
application(s)
detection
functional group
amine
carboxylic acid
thiol
storage temp.
2-8°C
SMILES string
N[C@@H](CCC(=O)N[C@@H](CS)C(=O)NCC(O)=O)C(O)=O
InChI
1S/C10H17N3O6S/c11-5(10(18)19)1-2-7(14)13-6(4-20)9(17)12-3-8(15)16/h5-6,20H,1-4,11H2,(H,12,17)(H,13,14)(H,15,16)(H,18,19)/t5-,6-/m0/s1
InChI key
RWSXRVCMGQZWBV-WDSKDSINSA-N
Looking for similar products? Visit Product Comparison Guide
Amino Acid Sequence
General description
Application
Biochem/physiol Actions
Other Notes
also commonly purchased with this product
comparable product
related product
Storage Class
11 - Combustible Solids
wgk_germany
WGK 2
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, type N95 (US)
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
Oxidative stress is mediated, in part, by reactive oxygen species produced by multiple cellular processes and controlled by cellular antioxidant mechanisms such as enzymatic scavengers or antioxidant modulators. Free radicals, such as reactive oxygen species, cause cellular damage via cellular.
Antioxidants protect biological systems from oxidative damage produced by oxygen-containing free radicals and from redoxactive transition metal ions such as iron, copper, and cadmium.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service