52650
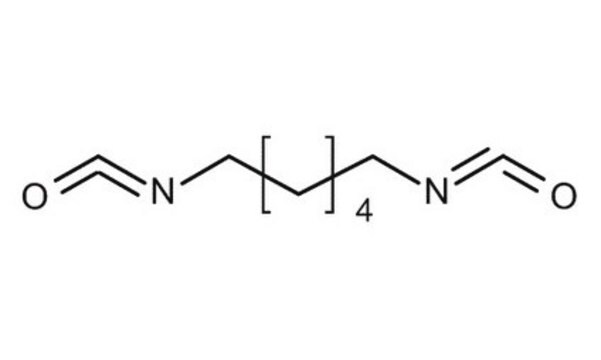
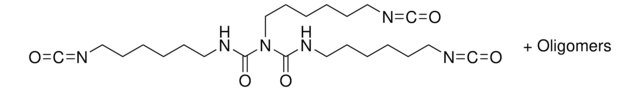
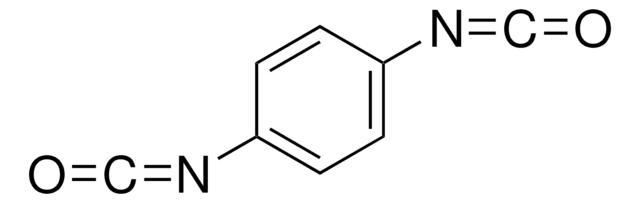
Hexamethylene diisocyanate
purum, ≥98.0% (GC)
Synonym(s):
1,6-Diisocyanatohexane
About This Item
Recommended Products
grade
purum
Quality Level
assay
≥98.0% (GC)
refractive index
n20/D 1.453
bp
82-85 °C/0.1 mmHg
density
1.047 g/mL at 20 °C (lit.)
SMILES string
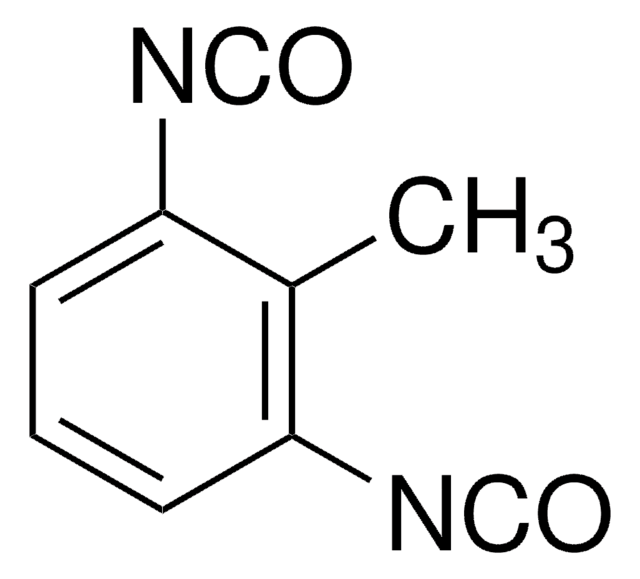
O=C=NCCCCCCN=C=O
InChI
1S/C8H12N2O2/c11-7-9-5-3-1-2-4-6-10-8-12/h1-6H2
InChI key
RRAMGCGOFNQTLD-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
General description
Application
- A crosslinker to crosslink the polyurethane chains in the triblock copolymer gate dielectric, which is then deposited on the substrate to fabricate low-voltage organic thin-film transistors.
- A precursor in the preparation of electroactive shape memory polyurethane/graphene nanocomposites. These materials are usually used as actuators, sensors, artificial muscles, smart devices, and microswitches.
- A crosslinker in conjunction with Pluronic F127, a nonionic surfactant, to synthesize a poly(lactic acid) (PLA)-based hydrogel for biomedical applications.
signalword
Danger
Hazard Classifications
Acute Tox. 1 Inhalation - Acute Tox. 4 Oral - Eye Dam. 1 - Resp. Sens. 1 - Skin Corr. 1C - Skin Sens. 1 - STOT SE 3
target_organs
Respiratory system
Storage Class
6.1A - Combustible acute toxic Cat. 1 and 2 / very toxic hazardous materials
wgk_germany
WGK 1
flash_point_f
266.0 °F - Pensky-Martens closed cup
flash_point_c
130 °C - Pensky-Martens closed cup
ppe
Eyeshields, Faceshields, Gloves, type ABEK (EN14387) respirator filter
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Protocols
HPLC Analysis of Isocyanates on Titan™ C18
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service