11684795910
Roche
In Situ Cell Death Detection Kit, Fluorescein
sufficient for ≤50 tests, suitable for detection
Synonym(s):
Fluorescein-12-UTP tetralithium salt, Fluorescein-5(6)-carboxamidocaproyl-(5-[3-aminoallyl]uridine 5′-triphosphate)
About This Item
Recommended Products
usage
sufficient for ≤50 tests
Quality Level
manufacturer/tradename
Roche
technique(s)
flow cytometry: suitable
application(s)
detection
storage temp.
−20°C
Related Categories
General description
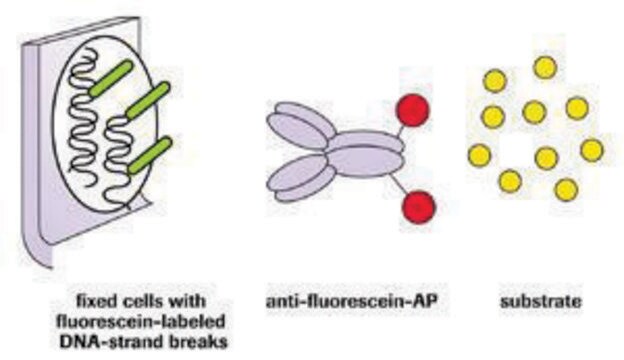
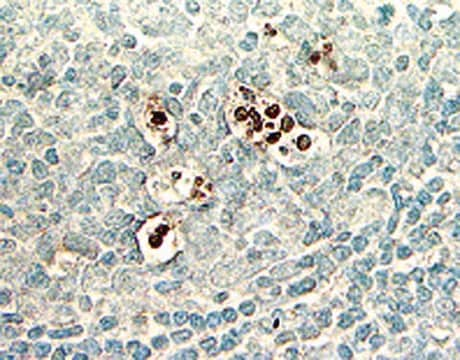
The hallmark of apoptosis is DNA degradation, which in early stages, is selective to the internucleosomal DNA linker regions. The DNA cleavage may yield double-stranded and single-stranded DNA breaks (nicks). Both types of breaks can be detected by labeling the free 3′-OH termini with modified nucleotides (e.g., biotin-dUTP, DIG-dUTP, fluorescein-dUTP) in an enzymatic reaction. The enzyme terminal deoxynucleotidyl transferase (TdT) catalyzes the template-independent polymerization of deoxyribonucleotides to the 3′-end of single- and double-stranded DNA. This method has also been termed TUNEL (TdT-mediated dUTP-X nick end labeling). Alternatively, free 3′-OH groups may be labeled using DNA polymerases by the template-dependent mechanism called nick translation. However, the TUNEL method is considered to be more sensitive and faster.
Specificity
Application
Examples are:
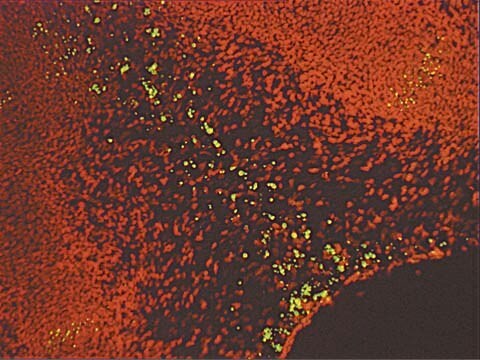
- Detection of individual apoptotic cells in frozen and formalin-fixed tissue sections in basic research
- Determination of sensitivity of malignant cells to drug-induced apoptosis in cancer research
- Typing of cells undergoing cell death in heterogeneous populations by double staining procedures
Features and Benefits
- Sensitive: The direct labeling procedure using fluorescein-dUTP reduces background labeling
- Fast: The use of fluorescein-dUTP allows analysis of the samples directly after the TUNEL reaction
- Convenient: No secondary detection system required
- Accurate: Identification of apoptosis at a molecular level (DNA-strand breaks) and identification of cells at the very early stages of apoptosis
Packaging
Preparation Note
Mix well to equilibrate components.
Other Notes
Kit Components Only
- Enzyme Solution (TdT)
- Label Solution (fluorescein-dUTP)
signalword
Danger
hcodes
Hazard Classifications
Aquatic Chronic 2 - Carc. 1B Inhalation
Storage Class
6.1D - Non-combustible acute toxic Cat.3 / toxic hazardous materials or hazardous materials causing chronic effects
wgk_germany
WGK 3
flash_point_f
does not flash
flash_point_c
does not flash
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
Cellular apoptosis assays to detect programmed cell death using Annexin V, Caspase and TUNEL DNA fragmentation assays.
Protocols
In Situ Cell Death Detection Kit, Fluorescein Protocol & Troubleshooting
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service