856177
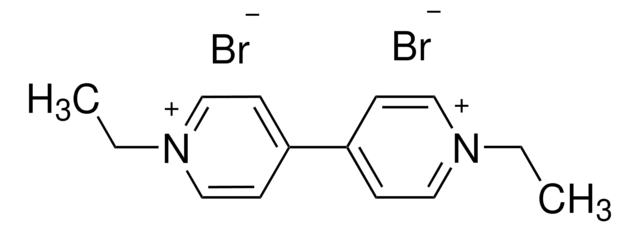
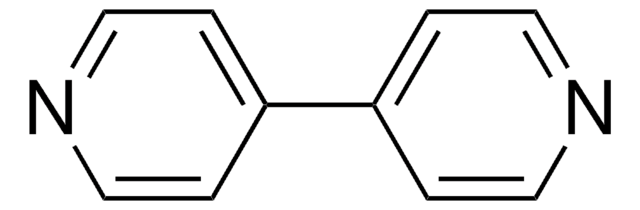
Methyl viologen dichloride hydrate
98%
Synonym(s):
1,1′-Dimethyl-4,4′-bipyridinium dichloride, Gramoxone, Paraquat dichloride
About This Item
Recommended Products
Quality Level
assay
98%
form
solid
mp
>300 °C (lit.)
SMILES string
[Cl-].[Cl-].[H]O[H].C[n+]1ccc(cc1)-c2cc[n+](C)cc2
InChI
1S/C12H14N2.2ClH.H2O/c1-13-7-3-11(4-8-13)12-5-9-14(2)10-6-12;;;/h3-10H,1-2H3;2*1H;1H2/q+2;;;/p-2
InChI key
RLGVBTFWTZJYIL-UHFFFAOYSA-L
Related Categories
General description
Application
It can also be used as a mediator in the synthesis of ultrasmall bimetallic PdAg nanocatalysts.
signalword
Danger
Hazard Classifications
Acute Tox. 2 Dermal - Acute Tox. 2 Inhalation - Acute Tox. 3 Oral - Aquatic Acute 1 - Aquatic Chronic 1 - Eye Irrit. 2 - Skin Irrit. 2 - STOT RE 1 - STOT SE 3
target_organs
Respiratory system
Storage Class
6.1A - Combustible acute toxic Cat. 1 and 2 / very toxic hazardous materials
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
dust mask type N95 (US), Eyeshields, Faceshields, Gloves, type P2 (EN 143) respirator cartridges
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Articles
Light-Emitting Polymers
Graphene has emerged as the new wonder material. Being only one atom thick and composed of carbon atoms arranged in a hexagonal honeycomb lattice structure, the interest in this material has exploded exponentially since 2004 when it was first isolated and identified using a very simple method.
Global Trade Item Number
| SKU | GTIN |
|---|---|
| 856177-1G | 4061835563982 |
| 856177-250MG | 4061835571949 |
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service