658235
Lithium dimethylaminoborohydride solution
1 M in THF
Synonym(s):
N-Methylmethanamine boron complex, Lithium (dimethylamino)trihydroborate, Lithium trihydro(N-methylmethanaminato)borate
About This Item
Recommended Products
reaction suitability
reagent type: reductant
concentration
1 M in THF
refractive index
n20/D 1.423
density
0.882 g/mL at 25 °C
storage temp.
2-8°C
SMILES string
[Li+].[BH3-]N(C)C
InChI
1S/C2H9BN.Li/c1-4(2)3;/h1-3H3;/q-1;+1
InChI key
CEDUMRZWZLVFKS-UHFFFAOYSA-N
Related Categories
General description
Application
Reactant for:
- B-H oxidative addition reactions
- Reduction and amination reactions
- Reduction of N-alkyl lactams
- Synthesis of tertiary amine-boranes
signalword
Danger
hcodes
Hazard Classifications
Acute Tox. 4 Oral - Carc. 2 - Eye Irrit. 2 - Flam. Liq. 2 - STOT SE 3
target_organs
Respiratory system
supp_hazards
Storage Class
4.3 - Hazardous materials which set free flammable gases upon contact with water
wgk_germany
WGK 3
flash_point_f
1.4 °F - closed cup
flash_point_c
-17 °C - closed cup
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
Lithium aminoborohydride (LAB) reagents are a new class of powerful and selective reagents developed in the laboratory of Professor Bakthan Singaram at the University of California, Santa Cruz.
Lithium aminoborohydride (LAB) reagents are a new class of powerful and selective reagents developed in the laboratory of Professor Bakthan Singaram at the University of California, Santa Cruz.
Related Content
here are many optically active organic compounds of biological and medicinal significance. For example, statine analogs, antibiotics, anesthetics, heterocyclic compounds, unusual amino acids, and insect pheromones all contain stereogenic centers. There is, therefore, a continuous need for new asymmetric methodology.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service



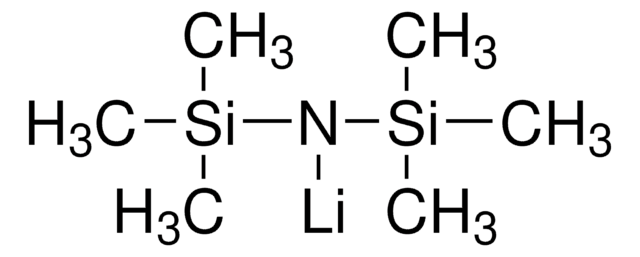
![[1,1′-Bis(diphenylphosphino)ferrocene]dichloropalladium(II), complex with dichloromethane](/deepweb/assets/sigmaaldrich/product/structures/825/986/4317978b-1256-4c82-ab74-6a6a3ef948b1/640/4317978b-1256-4c82-ab74-6a6a3ef948b1.png)


