212849
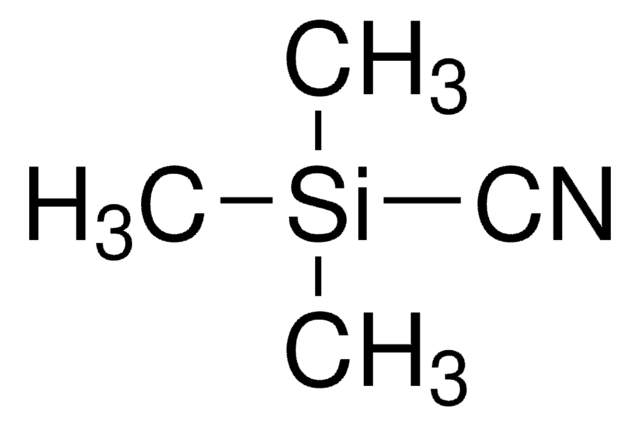
Trimethylsilyl cyanide
98%
Synonym(s):
Cyanotrimethylsilane, TMSCN
About This Item
Recommended Products
Quality Level
assay
98%
form
liquid
refractive index
n20/D 1.392 (lit.)
bp
114-117 °C (lit.)
mp
8-11 °C (lit.)
density
0.793 g/mL at 20 °C (lit.)
SMILES string
C[Si](C)(C)C#N
InChI
1S/C4H9NSi/c1-6(2,3)4-5/h1-3H3
InChI key
LEIMLDGFXIOXMT-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
Application
- for derivatization of complex metabolite mixtures by GC-MS
- as cyanide source in enantioselective organocatalytic Strecker-type reaction of aliphatic N,N-dialkylhydrazones
- as reagent for the cyanosilylation of aldehydes in near quantitative yield via catalysis by silylene-bridged rare earth oxide complexes
- in asymmetric cyanosilylation with chiral dinuclear Ti(IV) complexes
Packaging
signalword
Danger
Hazard Classifications
Acute Tox. 1 Dermal - Acute Tox. 2 Inhalation - Acute Tox. 2 Oral - Aquatic Acute 1 - Aquatic Chronic 1 - Flam. Liq. 2
supp_hazards
Storage Class
3 - Flammable liquids
wgk_germany
WGK 3
flash_point_f
33.8 °F - closed cup
flash_point_c
1 °C - closed cup
ppe
Eyeshields, Faceshields, Gloves, type ABEK (EN14387) respirator filter
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
We carry a large variety of electrophiles and nucleophiles that are widely used in C–C bond-forming reactions. This group of products contains many organometallic reagents as well as commonly-used alkylating and acylating reagents.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service