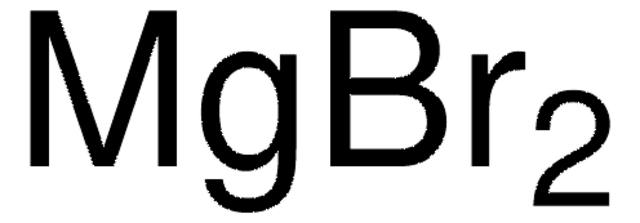208337
Magnesium chloride
powder, <200 μm
Synonym(s):
Magnogene
Sign Into View Organizational & Contract Pricing
All Photos(3)
About This Item
Linear Formula:
MgCl2
CAS Number:
Molecular Weight:
95.21
EC Number:
MDL number:
UNSPSC Code:
12352302
PubChem Substance ID:
NACRES:
NA.23
Recommended Products
form
powder
Quality Level
concentration
24.9-26.2% Mg (EDTA titration)
impurities
<5%
particle size
<200 μm
mp
714 °C (lit.)
density
2.32 g/mL at 25 °C (lit.)
SMILES string
Cl[Mg]Cl
InChI
1S/2ClH.Mg/h2*1H;/q;;+2/p-2
InChI key
TWRXJAOTZQYOKJ-UHFFFAOYSA-L
Looking for similar products? Visit Product Comparison Guide
Related Categories
General description
Also known as chloromagnestite, magnesium chloride is a colorless crystalline solid. It is very hygroscopic in nature. It is soluble in water and alcohol. It can be prepared either by heating hydrated magnesium chloride in the presence of hydrogel chloride or by directly heating magnesium ammonium chloride. Anhydrous magnesium chloride crystallizes from magnesium chloride hexahydrate as leaflets. It is electrically conductive. The crystal structure of MgCl2 is layer like. The structure is cubic close packing with alternate layers of octahedral holes occupied by Mg2+ ions.
Application
Used in salt baths, to maintain humidity, for the synthesis of poly (vinylpyrrolidone) and cellulose acetate nanofibres. It may have general applications in ceramic and textile industries.
Storage Class
13 - Non Combustible Solids
wgk_germany
WGK 1
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, type N95 (US)
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
The effect of temperature and humidity on electrospinning.
J. Mater. Sci., 44(5), 1357-1362 (2009)
Bert De Rybel et al.
Science (New York, N.Y.), 345(6197), 1255215-1255215 (2014-08-12)
Coordination of cell division and pattern formation is central to tissue and organ development, particularly in plants where walls prevent cell migration. Auxin and cytokinin are both critical for division and patterning, but it is unknown how these hormones converge
Elaine T Lim et al.
PLoS genetics, 10(7), e1004494-e1004494 (2014-08-01)
Exome sequencing studies in complex diseases are challenged by the allelic heterogeneity, large number and modest effect sizes of associated variants on disease risk and the presence of large numbers of neutral variants, even in phenotypically relevant genes. Isolated populations
Hideyuki Arita et al.
Brain tumor pathology, 32(1), 22-30 (2014-04-22)
Assessment of the mutational status of the isocitrate dehydrogenase 1/2 (IDH1/2) gene has become an integral part of the standard diagnostic procedure and, therefore, needs to be accurate. This may, however, be compromised by various factors including the method of
Stephen Q Wong et al.
Oncotarget, 6(2), 1115-1127 (2014-12-30)
Melanoma is often caused by mutations due to exposure to ultraviolet radiation. This study reports a recurrent somatic C > T change causing a P131L mutation in the RQCD1 (Required for Cell Differentiation1 Homolog) gene identified through whole exome sequencing
Protocols
Summary application report for analysis of moisture in Magnesium chloride
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service