134910
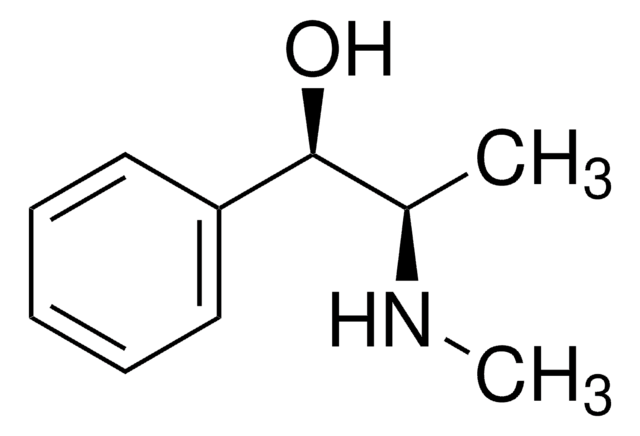
(1R,2S)-(−)-Ephedrine
98%
Synonym(s):
(−)-Ephedrine, (1R,2S)-(−)-α-(1-Methylaminoethyl)benzyl alcohol, (1R,2S)-(−)-2-Methylamino-1-phenyl-1-propanol, L-α-(1-Methylaminoethyl)benzyl alcohol
About This Item
Recommended Products
Quality Level
assay
98%
form
solid
optical activity
[α]21/D −41°, c = 5 in 1 M HCl
bp
255 °C (lit.)
mp
37-39 °C (lit.)
density
1.124 g/mL at 25 °C (lit.)
functional group
amine
hydroxyl
phenyl
storage temp.
2-8°C
SMILES string
CN[C@@H](C)[C@H](O)c1ccccc1
InChI
1S/C10H15NO/c1-8(11-2)10(12)9-6-4-3-5-7-9/h3-8,10-12H,1-2H3/t8-,10-/m0/s1
InChI key
KWGRBVOPPLSCSI-WPRPVWTQSA-N
General description
Application
signalword
Danger
hcodes
Hazard Classifications
Acute Tox. 4 Oral - Eye Dam. 1 - Skin Corr. 1B - Skin Sens. 1B
Storage Class
8A - Combustible corrosive hazardous materials
wgk_germany
WGK 1
flash_point_f
186.8 °F - closed cup
flash_point_c
86 °C - closed cup
ppe
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
Ultra-high performance liquid chromatographic (UHPLC) separation of ephedrine and its three structural analogs based on polar retention effect on graphite (PREG) mechanism using Supel™ Carbon LC column.
Chromatograms
application for HPLCapplication for HPLCOur team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service