推薦產品
等級
pharmaceutical primary standard
API 家族
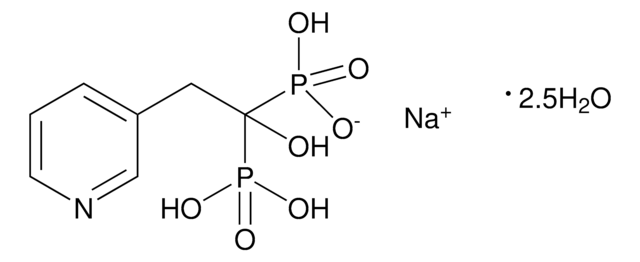
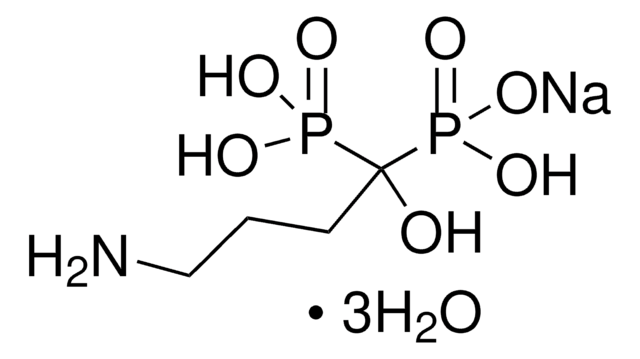
risedronate
製造商/商標名
USP
應用
pharmaceutical (small molecule)
形式
neat
SMILES 字串
OP(C(P(O)([O-])=O)(O)CC1=CC=CN=C1)(O)=O.[Na+]
InChI
1S/2C7H11NO7P2.2Na.5H2O/c2*9-7(16(10,11)12,17(13,14)15)4-6-2-1-3-8-5-6;;;;;;;/h2*1-3,5,9H,4H2,(H2,10,11,12)(H2,13,14,15);;;5*1H2/q;;2*+1;;;;;/p-2
InChI 密鑰
HYFDYHPNTXOPPO-UHFFFAOYSA-L
尋找類似的產品? 前往 產品比較指南
相關類別
一般說明
Risedronate sodium is a member of the pyridinyl class of bisphosphonates. It is mostly used as an antiresorptive agent. It can be used in treating and preventing postmenopausal and glucocorticoid-induced osteoporosis.
應用
Risedronate sodium USP reference standard, intended for use in specified quality tests and assays as specified in the USP compendia. Also, for use with USP monographs such as:
- Risedronate Sodium Tablets
- Risedronate Sodium Delayed-Release Tablets
分析報告
These products are for test and assay use only. They are not meant for administration to humans or animals and cannot be used to diagnose, treat, or cure diseases of any kind.
其他說明
Sales restrictions may apply.
相關產品
產品號碼
描述
訂價
訊號詞
Warning
危險分類
Acute Tox. 4 Oral - Eye Irrit. 2 - Repr. 2 - STOT SE 2
儲存類別代碼
11 - Combustible Solids
水污染物質分類(WGK)
WGK 3
閃點(°F)
Not applicable
閃點(°C)
Not applicable
客戶也查看了
CUNHA F.
American Journal of Surgery, 76(3), 244-260 (1948)
Eliska Vaculikova et al.
Molecules (Basel, Switzerland), 19(11), 17848-17861 (2014-11-07)
One approach for the enhancement of oral drug bioavailability is the technique of nanoparticle preparation. Risedronate sodium (Biopharmaceutical Classification System Class III) was chosen as a model compound with high water solubility and low intestinal permeability. Eighteen samples of risedronate
Yoshihisa Hirota et al.
PloS one, 10(4), e0125737-e0125737 (2015-04-16)
UbiA prenyltransferase domain-containing protein 1 (UBIAD1) plays a significant role in vitamin K2 (MK-4) synthesis. We investigated the enzymological properties of UBIAD1 using microsomal fractions from Sf9 cells expressing UBIAD1 by analysing MK-4 biosynthetic activity. With regard to UBIAD1 enzyme
Jin Woo Park et al.
Archives of pharmacal research, 37(12), 1560-1569 (2013-11-21)
Risedronate is widely used clinically to treat osteoporosis, Paget's disease, hypercalcemia, bone metastasis, and multiple myeloma. However, its oral efficacy is restricted due to its low bioavailability and severe gastrointestinal adverse effects. This study was designed to evaluate the effect
我們的科學家團隊在所有研究領域都有豐富的經驗,包括生命科學、材料科學、化學合成、色譜、分析等.
聯絡技術服務