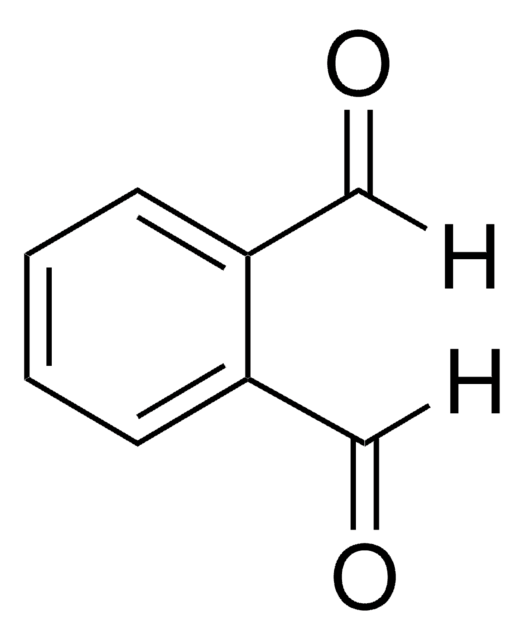
推薦產品
等級
certified reference material
pharmaceutical secondary standard
品質等級
API 家族
risedronate
CofA
current certificate can be downloaded
包裝
pkg of 50 mg
mp
161-163 °C (lit.)
應用
pharmaceutical
形式
neat
儲存溫度
2-30°C
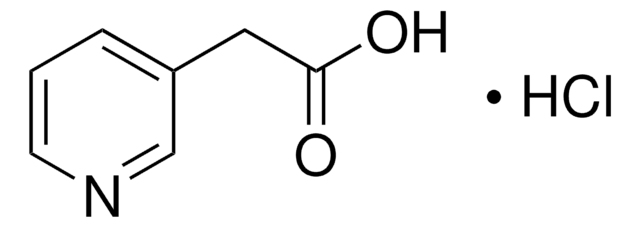
SMILES 字串
Cl[H].OC(=O)Cc1cccnc1
InChI
1S/C7H7NO2.ClH/c9-7(10)4-6-2-1-3-8-5-6;/h1-3,5H,4H2,(H,9,10);1H
InChI 密鑰
XVCCOEWNFXXUEV-UHFFFAOYSA-N
尋找類似的產品? 前往 產品比較指南
一般說明
Risedronate Impurity D is an impurity of risedronate, which is a new generation pyridinyl bisphosphonate, effectively used in the treatment of Paget′s disease of bone. It is also used for the treatment of skeletal disorders since it inhibits osteoclast-mediated bone resorption and modulates bone metabolism.
應用
Risedronate may be used as a pharmaceutical reference standard for the quantification of the analyte in pharmaceutical formulations using reversed-phase ion-pair high-performance liquid chromatographic method.
These Secondary Standards are qualified as Certified Reference Materials. These are suitable for use in several analytical applications including but not limited to pharma release testing, pharma method development for qualitative and quantitative analyses, food and beverage quality control testing, and other calibration requirements.
分析報告
These secondary standards offer multi-traceability to the USP, EP and BP primary standards, where they are available.
其他說明
This Certified Reference Material (CRM) is produced and certified in accordance with ISO 17034 and ISO/IEC 17025. All information regarding the use of this CRM can be found on the certificate of analysis.
腳註
To see an example of a Certificate of Analysis for this material enter LRAB8929 in the Documents slot below. This is an example certificate only and may not be the lot that you receive.
相關產品
產品號碼
描述
訂價
訊號詞
Warning
危險聲明
危險分類
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
標靶器官
Respiratory system
儲存類別代碼
11 - Combustible Solids
水污染物質分類(WGK)
WGK 3
閃點(°F)
Not applicable
閃點(°C)
Not applicable
Development and validation of a reversed-phase ion-pair high-performance liquid chromatographic method for the determination of risedronate in pharmaceutical preparations
Kyriakides D and Panderi I
Analytica Chimica Acta, 584(1), 153-159 (2007)
Stability indicating ion?pair HPLC method for the determination of risedronate in a commercial formulation
Aluoch A, et al.
Journal of Liquid Chromatography and Related Technologies, 27(17), 2799-2813 (2004)
我們的科學家團隊在所有研究領域都有豐富的經驗,包括生命科學、材料科學、化學合成、色譜、分析等.
聯絡技術服務